Clinical Pediatric Hematology-Oncology Volume 20ㆍNumber 1ㆍApril 2013 CASE REPORT
75
항암치료후 조기에 성장기형증후군이 발생한 중추신경계 비배아종성 생식세포종 1예
자오지아 장1,2ㆍ윤병규1ㆍ백희조1ㆍ박선주1ㆍ한동균1ㆍ박수민1ㆍ정태영3ㆍ이민철4ㆍ국 훈1
1화순전남대학교병원 소아청소년과, 2선진 인민병원 소아청소년과, 3화순전남대학교병원 신경외과, 4전남대학교 의과대학 병리학교실
Growing Teratoma Syndrome of the Pineal Gland Recognized Very Early during Chemotherapy in a Child with a Non-Germinomatous Germ-Cell Tumor
Zhaoxia Zhang, M.D.1,2, Byung Gyu Yoon, M.D.1, Hee Jo Baek, M.D.1, Sun Ju Park, M.D.1, Dong Kyun Han, M.D.1, Soo Min Park, M.D.1, Tae Young Jung, M.D.3, Min Cheol Lee, M.D.4 and Hoon Kook, M.D.1
1Department of Pediatrics, Chonnam National University Hwasun Hospital, Hwasun-gun, Korea,
2Department of Pediatrics, Shenzen People's Hospital, China, 3Department of Neurosurgery, Chonnam National University Hwasun Hospital, Hwasun-gun,
4Department of Pathology, Chonnam National University Medical School, Gwangju, Korea
The growing teratoma syndrome (GTS) is defined as the development of mature low-grade elements in the absence of a recurrent non-germinomatous germ-cell tumor (NGGCT) after partial response to multimodal treatment. It is uncommon and may occur in intracranial NGGCTs. Here, we report that a 7-year-old boy with intracranial NGGCT presented with precocious puberty and developed growing teratoma syndrome only 2 weeks after the first cycle of chemotherapy.
pISSN 2233-5250 / eISSN 2233-4580 Clin Pediatr Hematol Oncol 2013;20:75∼78
Received on April 1, 2013 Revised on April 11, 2013 Accepted on April 16, 2013
Corresponding author: Hoon Kook Department of Pediatrics, Chonnam National University Hwasun Hospital, Chonnam National University Medical School, 22, Seoyang-ro, Hwasun-eup, Hwasun-gun, Jeollanam-do 519-763, Korea Tel: +82-61-379-7696 Fax: +82-61-379-7697
E-mail: [email protected]
Key Words: Growing teratoma syndrome, Non-germinomatous germ cell tumors, Immature teratoma
Introduction
Germ cell tumors (GCT) usually develop from midline locations including the male and female gonads, retro- peritoneum, mediastinum, pineal gland and diencephalon [1]. Pineal GCTs account for 3-8% of intracranial tumors in children [2]. Three quarters of affected patients are between
6 and 24 years of age and are predominantly male adoles- cents (male/female ratio=4/1) [3]. GCTs are divided into germinomas (equivalent to gonadal seminomas and dysger- minomas) and teratomas and non-germinomatous germ cell tumors (NGGCTs). Logothetis et al. in 1982 first described the clinical presentation of a persistent or growing mass with normal or decreasing tumor markers during or after treatment of a NGGCT as the growing teratoma syndrome
Zhaoxia Zhang, et al
76 Vol. 20, No. 1, April 2013
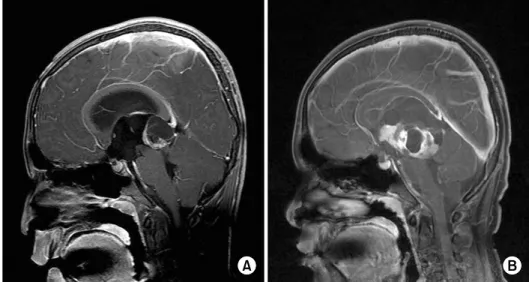
Fig. 1. (A) At diagnosis, MRI demon- strates a 3×2.7×2.5 cm cystic and solid mass in the pineal gland with obstructive hydrocephalus. (B) Two weeks after the first cycle of che- motherapy, T1-weighted MRI scan showed a marked increase in the tumor of 5.4×5.2×5.7 cm that consisted of multiseptated cystic and solid portions with hydrocephalus.
(GTS) [4]. GTS has often been reported in non-semi- nomatous extracranial GCTs [5-7]. It is most commonly ob- served at retroperitoneal disease sites, but has also been re- ported in the lungs, supraclavicular lymph nodes, and liver [8,9]. The frequency of the GTS in patients with metastatic NGGCT is between 1.9-7.6% [4,8].
The GTS in intracranial NGGCTs, however, is extremely rare. Only a few cases of intracranial GTS have been re- ported in the literature since the first report in 1991 [1,10-12]. Kim et al. reported that 6.5% (11/170) of patients with intracranial GCT had features of GTS. As all the cases of intracranial GTS originated from NGGCTs, the frequency of GTS in intracranial NGGCTs has been reported to be 21.2% (11/52) [13]. Recently, Kong et al. reported that the frequency of intracranial GTS was 11.5% in their series of 52 patients with intracranial GCTs [10]. Herein, a 7-year-old boy with precocious puberty had a very rapid increase of a mass within two weeks of discontinuing 1st cycle of che- motherapy despite falling tumor marker levels; this prompt- ed surgical resection leading to the diagnosis of the GTS.
Case Report
A 7-year-old boy presented with deepening of the voice and development of secondary sexual characteristics for six months; in addition, he had a persistent headache with vomiting for the past five days. The medical and family his- tory was noncontributory. The physical examination
showed Tanner stage 3-4 development of the pubic hair and penile growth with testicular enlargement. A complete blood count and the other biochemical tests were within normal ranges. The gonadotropin releasing hormone stim- ulation test was negative. The levels of serum α-fetoprotein (FP) and β-human chorionic gonadotropin (β-HCG) were 174.7 IU/mL (ref. 0-5.8 IU/mL), and 28 mIU/mL (ref. 0-5 mIU/mL), respectively. Magnetic resonance imaging (MRI) of the brain revealed a 3×2.7×2.5 cm, heterogeneously enhancing cystic and solid mass in the pineal gland with obstructive hydrocephalus (Fig. 1A). The spinal MRI and cytological examination of cerebrospinal fluid (CSF) re- vealed no evidence of tumor dissemination. The α -FP and β-HCG in CSF was elevated (34.9 IU/mL and 40 mIU/mL, respectively). The patient underwent endoscopic ven- triculotomy with biopsy. The pathologic findings were compatible with germinoma (Fig. 2A), but the patient diag- nosed with NGGCT by elevated tumor markers in serum and CSF. He was planned to be treated with the Korean Society Pediatric Neuro-Oncology-G082 protocol consisting of etoposide, carboplatin, cyclophosphamide and bleomy- cin followed by radiation therapy. Two weeks after the first cycle of chemotherapy, headache and vomiting developed.
The serum α -FP level decreased to 15.3 IU/mL and the β-HCG level became normal. However, a MRI showed marked increase in the size of the multiseptated cystic and solid mass (5.4×5.2×5.7 cm) in the pineal region with ob- structive hydrocephalus (Fig. 1B). A complete resection of
Growing Teratoma Syndrome in a CNS-NGGCT
Clin Pediatr Hematol Oncol 77
Fig. 2. (A) At diagnosis, histological findings are compatible with germinoma showing monomorphic cells with clear cytoplasm, round nuclei and prominent nucleoli (H&E, ×200). (B) At complete resection, microscopic findings at reveal both mature elements such as cartilage and glandular structures and immature elements such as primitive neuroepithelium (H&E, ×40).
the tumor was performed, and the pathological diagnosis was an immature teratoma, grade I (Fig. 2B). After surgery, the patient received the remaining three cycles of chemo- therapy and radiation therapy (craniospinal irradiation 36 Gy/primary site 18 Gy) in a complete response status.
Unfortunately, he relapsed at 21 months after off-treatment.
He received 6 cycles of chemotherapy, stereotactic surgery and autologous peripheral blood transplantation (APBSCT).
Now, ten months after APBSCT, he remains relapse-free.
Discussion
The GTS is a distinct clinicopathologic entity charac- terized by an enlarging mass during or after chemotherapy in the presence of normal or decreasing tumor marker lev- els [14]. The GTS or chemotherapeutic retroconversion are two terms used to describe the same disease entity. As the name implies, two etiologies have been proposed: selective destruction of the malignant components with the remain- ing chemo-resistant benign mature teratoma and pro- gressive growth (in favor of GTS) or malignant cell dediffer- entiation (in favor of chemotherapeutic retroconversion) [15]. The criteria for the diagnosis of the GTS include: (1) normalization of previously elevated serum tumor marker
levels; (2) increase in the tumor size during or after chemo- therapy; (3) the absence of any NGGCT component other than a teratoma at tumor resection [4].
Our patient developed an enlarging mass during chemo- therapy for an intracranial GCT. Although the possibility of tumor recurrence or treatment resistance should be consid- ered, the diagnosis of the GTS was made by histopathology showing only an immature teratoma component along with decrease of the tumor marker levels.
Recently, Kim et al., reported the median interval of GTS development from the start of chemotherapy was 12.8 months (range 3-56 months) [13]. In another report of 30 patients with the GTS, the median interval was 20 months (range 5-66 months) [6]. Our patient developed GTS with very short interval, 2 weeks after only one cycle of chemotherapy.
The prognosis of patients with the GTS is good, because it is a benign condition. However, the tumors associated with the GTS can grow very rapidly, even during treatment, causing deterioration of clinical signs and symptoms.
Surgical resection is the standard treatment for these pa- tients [7]. Recurrences have been reported in up to 83% of patients with incomplete resection compared to a 4% re- currence in patients with a complete resection [6]. Early de-
Zhaoxia Zhang, et al
78 Vol. 20, No. 1, April 2013
tection of growth of the mass allows for timely elective sur- gical intervention. Thus frequent surveillance with regular MRI follow-up should be required in patients with GCTs of the CNS during and after chemotherapy, when the pa- tient shows deterioration of the clinical status despite de- creasing tumor marker levels.
In conclusion, GTS may develop with very short interval after chemotherapy, seen as rapidly as 2 weeks in this case.
When a residual mass enlarges during or after treatment, GTS should be kept in mind in the differential diagnosis of tumor recurrence or progression.
References
1. Bi WL, Bannykh SI, Baehring J. The growing teratoma syn- drome after subtotal resection of an intracranial nongermino- matous germ cell tumor in an adult: case report. Neurosur- gery 2005;56:188.
2. Hoffman HJ, Otsubo H, Hendrick EB, et al. Intracranial germ-cell tumors in children. J Neurosurg 1991;74:545-51.
3. Matsutani M, Sano K, Takakura K, et al. Primary intracranial germ cell tumors: a clinical analysis of 153 histologically veri- fied cases. J Neurosurg 1997;86:446-55.
4. Logothetis CJ, Samuels ML, Trindade A, Johnson DE. The growing teratoma syndrome. Cancer 1982;50:1629-35.
5. Aide N, Comoz F, Sevin E. Enlarging residual mass after treat- ment of a nonseminomatous germ cell tumor: growing ter- atoma syndrome or cancer recurrence? J Clin Oncol 2007;25:
4494-6.
6. André F, Fizazi K, Culine S, et al. The growing teratoma syn-
drome: results of therapy and long-term follow-up of 33 patients. Eur J Cancer 2000;36:1389-94.
7. Schmoll HJ, Souchon R, Krege S, et al. European consensus on diagnosis and treatment of germ cell cancer: a report of the European Germ Cell Cancer Consensus Group (EGCCCG).
Ann Oncol 2004;15:1377-99.
8. Jeffery GM, Theaker JM, Lee AH, Blaquiere RM, Smart CJ, Mead GM. The growing teratoma syndrome. Br J Urol 1991;67:195- 202.
9. Choi JS, Lim YS, Lee OJ, Park HJ, Kim JY. A case of recurrent growing teratoma syndrome during chemotherapy for im- mature teratoma of the ovary. Clin Pediatr Hematol Oncol 2007;14:93-7.
10. Kong DS, Nam DH, Lee JI, Park K, Kim JH, Shin HJ.
Intracranial growing teratoma syndrome mimicking tumor re- lapse: a diagnostic dilemma. J Neurosurg Pediatr 2009;3:392-6.
11. O’Callaghan AM, Katapodis O, Ellison DW, Theaker JM, Mead GM. The growing teratoma syndrome in a nongerminomatous germ cell tumor of the pineal gland: a case report and review.
Cancer 1997;80:942-7.
12. Yagi K, Kageji T, Nagahiro S, Horiguchi H. Growing teratoma syndrome in a patient with a non-germinomatous germ cell tumor in the neurohypophysis-case report. Neurol Med Chir (Tokyo) 2004;44:33-7.
13. Kim CY, Choi JW, Lee JY, et al. Intracranial growing teratoma syndrome: clinical characteristics and treatment strategy. J Neurooncol 2011;101:109-15.
14. Jennings MT, Gelman R, Hochberg F. Intracranial germ-cell tumors: natural history and pathogenesis. J Neurosurg 1985;
63:155-67.
15. Amsalem H, Nadjari M, Prus D, Hiller N, Benshushan A.
Growing teratoma syndrome vs chemotherapeutic retroconver- sion: case report and review of the literature. Gynecol Oncol 2004;92:357-60.