Introduction
Metabolicsyndrome
(
MS)
is characterized bycentral obesity,
dyslipidemia,
elevated blood pressure,
and highfasting glucose levels.
The MShasbeenrecognizedasapotentatherogenicstate and an establishedrisk factor ofcardiovascular diseaseand all-
cause mortality.
1) Before onset of cardiovascular disease,
the subclinical atherosclerosis is often present.
Therefore,
for the prevention of cardiovascular disease,
it is critical to identifyasymptomaticindividualswithatherosclerotic diseaseandmodify metabolicriskfactorsatthesubclinicalstage
.
Arterial stiffness is commonly used atherosclerotic markers and is recognized as an independent predictor for cardiovascular disease and cardiovascular mortality
.
2, 3) Increasedarterialstiffnesshasbeenalsodemonstratedinsubjects with MS.
4)With regardto arterialstiffness,
most ofthe studies investigating the impact of MS haveundertaken with pulse-
wavevelocity(
PWV).
4-9)However,
PWVishighlycorrelatedwithGender–dependent association between the metabolic syndrome and arterial stiffness in Korean adults.
최형화, 한건희, 한지혜*
을지의과대학교 노원을지병원 가정의학과
Gender-dependent association between the metabolic syndrome and arterial stiffness in Korean adults.
Hyung-Hwa Choi, Kun-Hee Han, Jee-Hye Han*
Department of Family medicine, Eulji General Hospital, College of Medicine, Eulji University, Seoul, Korea,
Background: The aim of this study was to determine the association between metabolic syndrome(MS) and arterial stiffness in both genders through assessed assessment by of cardio- ankle vascular index(CAVI) as an indicator of arterial stiffness and whether the association differs by gender.
Methods: A total of 1,393 Korean adults(mean age: 46.0 years) were included. MS was defined using the 2005 revised National Cholesterol Education Program-Adult Treatment Panel III criteria with modification of waist circumference cutoff points by the Korean society of the study of obesity. Arterial stiffness was assessed by measurement of CAVI, and high CAVI was defined as ≥ 8.0.
Results: Age group-specific average CAVI became higher in both genders as their age advanced. CAVI was significantly higher in men than in women in all age groups except for the group aged 60~79 years. CAVI was significantly associated with all of the cardiovascular risk factors except homocysteine in women, but only associated with age and HbA1c in men. CAVI increased with the increased number of MS components in women but not in men. CAVI with MS were significantly higher in women than in men(7.67±1.24 vs 7.34±0.85, P=0.025).
High CAVI group was significantly associated with MS [ORs(95% CI): 1.53(1.00~2.33) in men, 6.24(3.65~10.67) in women, respectively]. After adjustment for age and smoking status, only women showed significant association between high CAVI group and MS [OR(95%CI): 2.36(1.18~4.71)].
Conclusion: This study suggests that the MS is associated with arterial stiffness as assessed by CAVI, which is more pronounced in women than in menand this association is more pronounced in women than men.
Keywords: Cardio-ankle vascular index, Metabolic syndrome, Arterial stiffness
Received September 8, 2013 Accepted 8, 2015 Corresponding Author Jee-Hye Han
Tel: +82-2-970-8515, Fax: +82-2-970-8862 E-mail: [email protected]
Copyright © 2015 The Korean Academy of Family Medicine
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Hyung-Hwa Choi, et al. Gender-dependent association between the metabolic syndrome and arterial stiffness in Korean adults.
Korean Journal of Family Practice
KJFP
blood pressure
(
BP)
evenafter age adjustment.
10)Cardio-
ankle vascularindex(
CAVI)
wasdevelopedasarelativelynewindexof arterialstiffness whichis lessinfluenced byBP.
11)CAVIstudies showreproducibleandBP-
independentriskpredictability,
thus suggestingitssuperiorityoverPWV.
10, 12)Several observational studies suggest that theMS increases the cardiovascular morbidity in women to a greater extent
than in men
.
13-14) And previous studies have also suggestedthat thegender
-
dependent association between the MS and atherosclerosis.
6, 15)The aim of this study was to determine the association betweenMSandarterialstiffnessassessedbyCAVIandwhether theassociationdiffersbygender
.
Methods
Study subjects
The study included 1393 subjects
,
774(
55.
6%)
men and 619(
44.
4%)
women,
between 20~
79 years of age(
mean age,
46.
0 years).
Study participants visited the health promotion center in general hospital for medical checkup from January 2010 to February 2013.
Subjects with stroke,
atherosclerotic cardiovascular disease,
cancer,
thyroid disorders,
apparent hepatic or renaldisease,
anemia,
or autoimmunedisease,
were excluded from the study.
Demographic data werecollectedby self-
reportedquestionnaires.
Measurements of CAVI
CAVIwasmeasurednoninvasivelybytheuseofaVaseraVS
-
1500(
FukudaDenshi Co.
Ltd.,
Tokyo,
Japan)
vascular screening device.
CAVIwasobtainedbysubstitutingthestiffnessparameter β into an equation for determining vascular elasticity and PWV.
The stiffnessβwasdefinedastheBP-
independentlocal stiffnessofbloodvessels,
basedonachangeinvasculardiameter correspondingtoarterialpressurevariance.
CAVIwas calculatedbythe following formula
:
CAVI=
a[
2ρ/
ΔP×(
InPs/
Pd)×
PWV2]+
b;
where a and b are constants;
ρ is blood density;
Psis systolicBP(
SBP);
Pd is diastolic BP(
DBP)
and ΔP isdifference in SBPand DBP.
The detailedprinciples underlyingCAVIhavebeenreportedpreviously.
11, 16)Cuffs to measure CAVI were wrapped around both upper armsandankleswhileparticipantswereinsupineposition
.
Afterat least 5minutes ofbed rest
,
measurements wereperformed.
Electrocardiography,
phonocardiography,
and pressures and waveformsofbrachialandanklearteriesweremeasured.
Allthe measurement andcalculationsareperformed automatically.
The resultswerecalculatedseparatelyfortheleftandtherightsideof thebody.
TheaveragevalueoftherightandleftCAVIwasused fortheanalysis.
Diagnostic criteria of MS
MS was defined according to the 2005 revised National Cholesterol Education Program
-
Adult Treatment Panel III(
NCEP ATP III)
criteria with modification of waist cir-
cumference cutoff points by theKorean Society of the Study ofObesity.
1, 17)Participantshaving3ofthefollowingmetabolic components were defined as having MS:
1)
increased waist circumference to≥
90cm inmen or≥
80cm in women;
2)
elevated BPto≥
130/
85mmHgorreceivinganti-
hypertensive medication;
3)
elevated serumtriglycerides level to≥
150mg/
dl or on drug treatment forelevated triglycerides;
4)
reduced serum high-
density lipoprotein-
cholesterol(
HDL-
C)
to<40mg
/
dl in men and <50mg/
dl in women or on drug treatment for reduced HDL-
C;
5)
elevated fasting glucose level(
FPG)
to≥
100mg/
dlorreceivinganti-
diabeticmedication.
Laboratory and physical measurementsMedical history was recorded by self
-
administered ques-
tionnaires.
Themeasurementsofbody weight,
height,
andwaist circumference weredone.
BPwas measured twice inthe right upper arm in thesitting positionaftera 10 minutesrest using an automatic sphygmomanometer.
Body mass index(
BMI)
was calculated as body weight(
kg)
divided by the square of body height(
m).
Venous blood wascollected in the morning after overnightfasting.
The blood samplewas analyzed forthe followingparametersusingstandardtechniques:
totalcholesterol,
triglyceride,
low densitylipoprotein-
cholesterol(
LDL-
C),
high-
density lipoprotein-
cholesterol(
HDL-
C),
FPG,
hemoglobin A1c(
HbA1c),
fasting insulin,
high-
sensitive C-
reactive protein(
hs-
CRP),
apolipoprotein B,
and homocystein.
The homeostasismodel assessment ofinsulin resistance(
HOMA-
IR)
was calculated using thefollowing formula:
fasting insulin(
μU/
ml
)×
fastingplasmaglucose(
mmol/
L)/
22.
5.
Statistical analysisData was presented as the mean
±
standard deviation(
SD)
and p<0.
05was consideredstatistically significant.
Allanalyses were performedseparatelyformenandwomen.
Thedifferences in continuous or categoricalvariables between the twogroups were examinedby unpaired Student’
s t-
test orchi-
square test.
One-
wayanalysis of variance(
ANOVA)
was usedto compare CAVI accordingtoagegroups.
Pearson’
scorrelationcoefficients were usedto investigate the correlationof CAVI withvarious cardiovascular riskfactors.
CAVI werestratified by thenumber ofcomponentsofMS(
0,
1,
2,
3-
5).
One-
wayANOVAwasused toevaluatethetrendofCAVIwiththeincreasingnumberofMS components.
Logisticregressionanalysiswasusedtoestimatethe odds ratios ofhigh CAVIassociated with MSandits individualcomponents
.
The predefined cutoff valueof high CAVI was determinedtobe8.
0accordingtopreviousstudy.
19) Dataanalyses wereperformedbySPSSsoftwareforwindows(
StatisticalProduct andServiceSolutions,
version12.
0)
Results
The demographic data and studyparametersfor allsubjects are summarizedinTable 1
.
Menhad higherCAVI,
BMI,
WC,
SBP,
DBP,
totalcholesterol,
triglyceride,
LDL-
C,
fastingplasma glucose,
HbA1c,
HOMA-
IR,
hs-
CRP,
apolipoprotein B,
homocystein,
andtheproportionofcurrentandformersmokers thanwomen.
WomenhadhigherHDL-
Cthanmen.
TheNCEP ATPIIIcriteriaidentified256of1393participants(
18.
4%),
171of 774men(
22.
1%)
and85of619women(
13.
7%)
withMS.
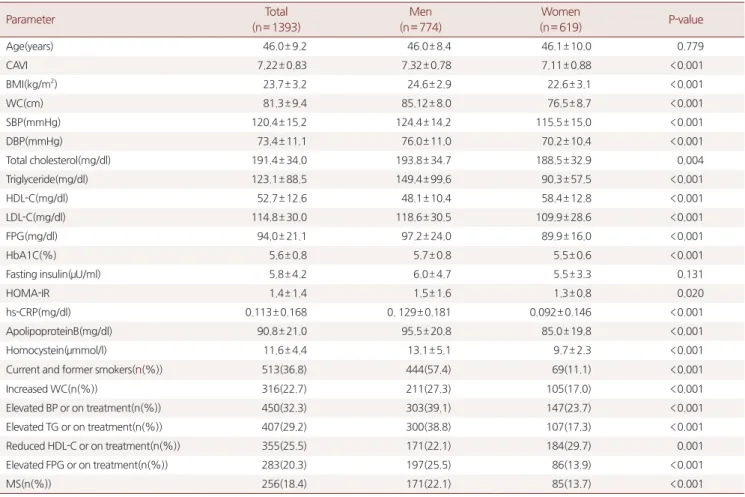
Figure 1 representsmeans andSD of the CAVI byage and gender
.
Agegroup-
specificaverageCAVIbecamehigherinbothTable 1. General characteristics
Parameter Total
(n= 1393)
Men (n= 774)
Women
(n= 619) P-value
Age(years) 46.0± 9.2 46.0± 8.4 46.1± 10.0 0.779
CAVI 7.22± 0.83 7.32± 0.78 7.11± 0.88 <0.001
BMI(kg/m2) 23.7± 3.2 24.6± 2.9 22.6± 3.1 <0.001
WC(cm) 81.3± 9.4 85.12± 8.0 76.5± 8.7 <0.001
SBP(mmHg) 120.4± 15.2 124.4± 14.2 115.5± 15.0 <0.001
DBP(mmHg) 73.4± 11.1 76.0± 11.0 70.2± 10.4 <0.001
Total cholesterol(mg/dl) 191.4± 34.0 193.8± 34.7 188.5± 32.9 0.004
Triglyceride(mg/dl) 123.1± 88.5 149.4± 99.6 90.3± 57.5 <0.001
HDL-C(mg/dl) 52.7± 12.6 48.1± 10.4 58.4± 12.8 <0.001
LDL-C(mg/dl) 114.8± 30.0 118.6± 30.5 109.9± 28.6 <0.001
FPG(mg/dl) 94.0± 21.1 97.2± 24.0 89.9± 16.0 <0.001
HbA1C(%) 5.6± 0.8 5.7± 0.8 5.5± 0.6 <0.001
Fasting insulin(μU/ml) 5.8± 4.2 6.0± 4.7 5.5± 3.3 0.131
HOMA-IR 1.4± 1.4 1.5± 1.6 1.3± 0.8 0.020
hs-CRP(mg/dl) 0.113± 0.168 0. 129± 0.181 0.092± 0.146 <0.001
ApolipoproteinB(mg/dl) 90.8± 21.0 95.5± 20.8 85.0± 19.8 <0.001
Homocystein(μmmol/l) 11.6± 4.4 13.1± 5.1 9.7± 2.3 <0.001
Current and former smokers(n(%)) 513(36.8) 444(57.4) 69(11.1) <0.001
Increased WC(n(%)) 316(22.7) 211(27.3) 105(17.0) <0.001
Elevated BP or on treatment(n(%)) 450(32.3) 303(39.1) 147(23.7) <0.001
Elevated TG or on treatment(n(%)) 407(29.2) 300(38.8) 107(17.3) <0.001
Reduced HDL-C or on treatment(n(%)) 355(25.5) 171(22.1) 184(29.7) 0.001
Elevated FPG or on treatment(n(%)) 283(20.3) 197(25.5) 86(13.9) <0.001
MS(n(%)) 256(18.4) 171(22.1) 85(13.7) <0.001
Values are presented as mean±standard deviation or number(%). Analyzed by Student’s t-test or chi-squared test.
Abbreviations: CAVI, cardio-ankle vascular index; BMI, body mass index; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein-cholesterol; LDL-C, low-density lipoprotein-cholesterol; FPG, fasting plasma glucose; HbA1C, hemoglobin A1c; HOMA-IR, homeostatic model assessment-insulin resistance; hs-CRP, high-sensitive C-reactive protein; BP, blood pressure; MS, metabolic syndrome.
Hyung-Hwa Choi, et al. Gender-dependent association between the metabolic syndrome and arterial stiffness in Korean adults.
Korean Journal of Family Practice
KJFP
gendersastheirageadvanced
.
CAVIsweresignificantlyhigherin menthaninwomeninallagegroupsexceptforthegroupaged 60~
79years.
In women
,
CAVI was significantly correlated with all of the cardiovascular risk factors except homocystein.
In men,
CAVI wasonly correlatedwithage andHbA1c(
Table2).
CAVI increasedwithanincreasednumberofMScomponentsintotal subjectsandwomenbutnotinmen(
Table3).
CAVIwithoutMS weresignificantlyhigherinmenthanwomen(
7.
32±
0.
78vs7.
11±
0.
88,
P<0.
001).
However,
CAVIwithMSweresignificantlyhigher inwomenthaninmen(
7.
67±
1.
24vs7.
34±
0.
85,
P=
0.
025).
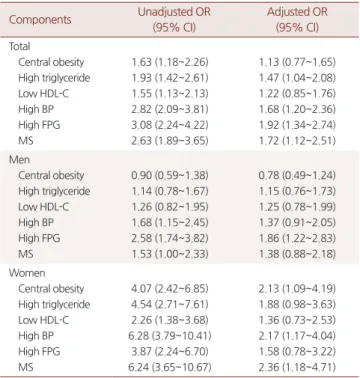
As shown in Table 4
,
high CAVI group was significantly associated withMSinmenandwomen.
TheORs(
95%
CI)
were 1.
53(
1.
00~
2.
33)
in men and 6.
24(
3.
65-
10.
67),
respectively.
After adjustment for age and smoking status
,
only women showedsignificantassociationbetweenhighCAVIgroupandMS[
adjustedOR(
95%
CI):
2.
36(
1.
18-
4.
71)].
Discussions
ThisstudyshowsCAVIincreasedwithagelinearlyfrom20to 70yearsinmenandwomen
.
Shiraietal.20) demonstratedadirect association betweenCAVI andAge.
CAVIisknowntocorrelate positivelywithage.
16)Agingenhancesthedegenerationofelastic fibersand isassociated withprogressivestiffeningofthearterial wall.
21)Inthisstudy
,
CAVI ofmenis higherthanthatofwomen in all agegroupsexcept theoldest group(
60~
79 years).
Namekata etal.
16)reported thatthe sexdifference inCAVI disappearedin olderindividuals(≥
70years).
Andsimilarresultswerefound inTable 2. Correlations between cardio-ankle vascular index and various cardiovascular risk factors
Variables Total Men Women
Age(years) 0.523** 0.498** 0.557**
BMI(kg/m2) 0.123** 0.042 0.138**
WC(cm) 0.142** 0.041 0.148**
SBP(mmHg) 0.104** 0.006 0.138**
DBP(mmHg) 0.078** 0.015 0.085*
Total cholesterol(mg/dl) 0.057* -0.019 0.126**
Triglyceride(mg/dl) 0.094** 0.029 0.116**
HDL-C(mg/dl) -0.120** -0.050 -0.098*
LDL-C(mg/dl) 0.062* -0.033 0.137**
FPG(mg/dl) 0.096** 0.065 0.100*
HbA1c(%) 0.134** 0.086* 0.182**
hs-CRP(mg/dl) 0.055* -0.008 0.115**
Apolipoprotein B(mg/dl) 0.125** 0.027 0.184**
Homocystein(μmmol/l) 0.051 0.015 0.085
* P-value <0.05 and ** P-value <0.01 by Pearson correlation analysis.
Abbereviations as in Table 1.
Values are presented as mean±standard deviation
* P-value <0.001 compared age-group specific CAVI within total, men, or women by one-way ANOVA and trend test;
† P-value <0.05 by Student’s t-test, men versus women.
Figure 1. Comparison of cardio-ankle vascular index by age and gender
Table 3. Cardio-ankle vascular index according to the number of components of metabolic syndrome. Subjects Number of metabolic components
Subjects Number of metabolic components
P-value P-trend
0 1 2 ≥ 3
Total No.
CAVI (SD)
497 7.03± 0.65
359 7.22± 0.76
281 7.37 ± 0.93
256
7.45± 1.01 <0.001 <0.001
Men No.
CAVI (SD)
207 7.25± 0.65
197 7.29± 0.74
199 7.41± 0.87
171
7.34± 0.85 0.211 0.136
Women No.
CAVI (SD)
290 6.87± 0.61
162 7.14± 0.78
82 7.28± 1.07
85
7.67± 1.24 <0.001 <0.001 Values are presented as mean±standard deviation. P-value and P-value for trend by ANOVA
Abbreviations: CAVI, cardio-ankle vascular index.
studies using PWV
.
5)Menopausemay augment theage-
related increase in arterial stiffness.
Estrogen deficiency during the postmenopausalphaseisknowntohaveanunfavorableinfluence on conventionalatherosclerotic riskfactor,
therebyaccelerating the progression of atherosclerosis and influencing arterial stiffness.
8)The presentstudyshowsgenderdifferencesintheassociation of theMS with arterialstiffness
.
The MS hasmore deleterious impact on arterial stiffness in women with MS rather than in men.
In women,
CAVI showed closer relationship with cardiovascular risk factors compared to men.
CAVI gradually increasedwiththenumberofMScomponentsonlyinwomen.
The gender specific effectsof MSon arterial stiffnessare in agreement with previous studies using PWV
,
a conventional measure ofarterial stiffness.
Ferreira et al.
7)found that PWV increased with increasing traits of MS only in women.
PWV showedcloserrelationshipwithcardiovascularriskfactorsofthe MSinwomen comparedtomenin Korean.
9)A recentstudy in China alsofoundthattheMSand itscomponentsaffectarterial stiffness more severely in women than in men.
5)And several studies suggest that the risk of cardiovascular morbidity and mortality associated with MSand diabetes is higherin womenthan in men
.
Thepresent findings that the MS has more deleterious impact on arterial stiffness inwomen thanin men mayexplainthehighercardiovascularriskinwomen.
Sexhormonesmayhaveamodulatingroleonarterialstiffness
.
It iswell known that theendogenous estrogen has protective effectsonthevasculatureinwomen.
22)Insulinresistanceislinked to lower estrogen concentrations and hyperandrogenicity.
23) Insulin resistance and itshormonalchanges in MSmay reverse thefavorableeffectsoffemale sexhormone.
FSHconcentrations correlate strongly with decline in central arterial function.
24) And genetic mechanisms mightrelate to thegender differences supported by the identification of sex-
specific quantitative geneticlocifortheMSriskcomponents.
25-26)PWV is a non
-
invasive method for predicting cardio-
vascular morbidity27)and mortality.
3)However,
PWV hasbeen known to besignificantly affected by many factors,
especially BP.
11)The CAVI wasdeveloped to obtain aBP-
independent arterial stiffness index.
11,
20)The index is calculated from the heart-
anklePWV,
adjustedforBPbasedon stiffnessparameter β.
Stiffness parameter βrepresents the BPchange required to expand the diameter of the artery.
Therefore,
arterial stiffness indexmeasuredbyCAVIisnotaffectedbyBPandquantitatively reflectsarteriosclerosis ofthe aorta,
femoral andtibial arteries.
High CAVI may indicate that the atherosclerotic process is already well established.
And it has been demonstrated that CAVIcorrelates positivelywith increased carotidintima-
media thickness(
IMT)
andapresenceofcarotidplaque.
12)Studies have found that CAVI was strongly associated with the presence of severecoronaryarteriosclerosis2)andcouldbeusedasascreening toolforarteriosclerosis.
28)Therearesomelimitationsinthisstudy
.
Thestudywascross-
sectional and did not include variouslifestyle factorssuch as alcoholconsumptionandexercise.
Thestrengthofthisstudyisthesex
-
specificassessmentforthe association of CAVIwith MS,
which wasrarely reported.
Our results suggest that MShad a strongerassociation with arterial stiffnessinwomenthaninmen.
In conclusion
,
MS had a stronger association with arterial stiffness assessed by CAVI in women than in men.
Gender differences cannot be explained sufficiently by available data,
whichimplicatestheneedforfurtherinvestigationsonthistopic.
Table 4. Odds ratios of high CAVI (CAVI≥8) according to metabolic syndrome and its components
Components Unadjusted OR (95% CI)
Adjusted OR (95% CI) Total
Central obesity High triglyceride Low HDL-C High BP High FPG MS
1.63 (1.18~2.26) 1.93 (1.42~2.61) 1.55 (1.13~2.13) 2.82 (2.09~3.81) 3.08 (2.24~4.22) 2.63 (1.89~3.65)
1.13 (0.77~1.65) 1.47 (1.04~2.08) 1.22 (0.85~1.76) 1.68 (1.20~2.36) 1.92 (1.34~2.74) 1.72 (1.12~2.51) Men
Central obesity High triglyceride Low HDL-C High BP High FPG MS
0.90 (0.59~1.38) 1.14 (0.78~1.67) 1.26 (0.82~1.95) 1.68 (1.15~2.45) 2.58 (1.74~3.82) 1.53 (1.00~2.33)
0.78 (0.49~1.24) 1.15 (0.76~1.73) 1.25 (0.78~1.99) 1.37 (0.91~2.05) 1.86 (1.22~2.83) 1.38 (0.88~2.18) Women
Central obesity High triglyceride Low HDL-C High BP High FPG MS
4.07 (2.42~6.85) 4.54 (2.71~7.61) 2.26 (1.38~3.68) 6.28 (3.79~10.41)
3.87 (2.24~6.70) 6.24 (3.65~10.67)
2.13 (1.09~4.19) 1.88 (0.98~3.63) 1.36 (0.73~2.53) 2.17 (1.17~4.04) 1.58 (0.78~3.22) 2.36 (1.18~4.71) Analyzed by logistic regression analysis adjusted for age and smoking status.
Abbreviations: OR, Odds ratio; CI, confidence interval; other abbreviations are the same as in Table 1.
Hyung-Hwa Choi, et al. Gender-dependent association between the metabolic syndrome and arterial stiffness in Korean adults.
Korean Journal of Family Practice
KJFP
요 약
연구배경: 이 연구는 대사증후군과
cardio-
ankle vascular index(
CAVI)에 의해 측정된 동맥 경직도의 관계를 조사하였다.
방법: 총
1.
393명의 한국인(평균
46.
2세)을 대상으로 하였다. 대 사증후군은
2005년에 개정된
National Cholesterol Education Program-
AdultTreatmentPanelIII의 기준과 한국비만학회의 복
부비만에 대한 한국인의 기준으로 정의하였다.
CAVI를 측정하여
8
.
0을 기준으로 고
CAVI군과 저
CAVI군으로 나누었다.
결과: 남성 및 여성에서 연령이 증가할수록
CAVI가 높았다.
60세 미만의 연령군에서 남성이 여성보다 유의하게
CAVI가 높았다. 여
성에서
CAVI는 호모시스테인을 제외한 심혈관질환 위험인자와 유
의한 상관관계를 보였으나 남성에서는 연령과 당화혈색소만이 유 의한 상관관계를 보였다. 여성에서 대사증후군 구성요소의 수가 증 가할수록
CAVI가 유의하게 증가하였으나, 남성에서는 유의하지 않 았다. 대사증후군에서의
CAVI값은 여성이 남성보다 높았다(
7.
67±
1.
24vs7.
34±
0.
85,
P=
0.
025). 고
CAVI군과 대사증후군의 교 차비는 남성에서
1.
53(
95%
CI:
1.
00~
2.
33), 여성에서
6.
24(
95%
CI:
3.
65~
10.
67)로 유의하였으나, 연령과 흡연력을 보정한 후에는 여성에서만 유의한 교차비(
2.
36,
95%
CI:
1.
18~
4.
71)를 보였다.
결론: 대사증후군은
CAVI로 측정된 동맥 경직도와 연관성이 있었 으며, 여성에서 남성보다 더 유의하였다.
중심단어:
Cardio-
anklevascularindex, 대사증후군, 동맥 경직도.
REFERENCES
1. Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 2005;112:2735-52.
2. Nakamura K, Tomaru T, Yamamura S, Miyashita Y, Shirai K, Noike H. Cardio-ankle vascular index is a candidate predictor of coronary atherosclerosis. Circ J 2008;72:598-604.
3. Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, Guize L, et al. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 2001;37:1236-41.
4. Satoh H, Kishi R, Tsutsui H. Metabolic syndrome is a significant and independent risk factor for increased arterial stiffness in Japanese subjects. Hypertens Res 2009;32:1067-71.
5. Weng C, Yuan H, Yang K, Tang X, Huang Z, Huang L, et al.
Gender-specific association between the metabolic syndrome and arterial stiffness in 8,300 subjects. Am J Med Sci 2013;346:289-94.
6. Lin HF, Liu CK, Liao YC, Lin RT, Chen CS, Juo SH. The risk of the metabolic syndrome on carotid thickness and stiffness: sex and age specific effects. Atherosclerosis 2010;210:155-9.
7. Ferreira I, Boreham CA, Twisk JW, Gallagher AM, Young IS, Murray LJ, et al. Clustering of metabolic syndrome risk factors and arterial stiffness in young adults: the Northern Ireland Young Hearts Project. J Hypertens 2007;25:1009-20.
8. Zaydun G, Tomiyama H, Hashimoto H, Arai T, Koji Y, Yambe M, et al. Menopause is an independent factor augmenting the age- related increase in arterial stiffness in the early postmenopausal phase. Atherosclerosis 2006;184:137-42.
9. Choi KM, Lee KW, Seo JA, Oh JH, Kim SG, Kim NH, et al.
Relationship between brachial-ankle pulse wave velocity and cardiovascular risk factors of the metabolic syndrome. Diabetes Res Clin Pract 2004;66:57-61.
10. Satoh N, Shimatsu A, Kato Y, Araki R, Koyama K, Okajima T, et al. Evaluation of the cardio-ankle vascular index, a new indicator of arterial stiffness independent of blood pressure, in obesity and metabolic syndrome. Hypertens Res 2008;31:1921-30.
11. Shirai K, Utino J, Otsuka K, Takata M. A novel blood pressure- independent arterial wall stiffness parameter; cardio-ankle vascular index(CAVI). J Atheroscler Thromb 2006;13:101-7.
12. Izuhara M, Shioji K, Kadota S, Baba O, Takeuchi Y, Uegaito T, et al.
Relationship of cardio-ankle vascular index(CAVI) to carotid and coronary arteriosclerosis. Circ J 2008;72:1762-7.
13. Hu G, Group DS. Gender difference in all-cause and cardiovascular mortality related to hyperglycaemia and newly-diagnosed diabetes.
Diabetologia 2003;46:608-17.
14. McNeill AM, Rosamond WD, Girman CJ, Golden SH, Schmidt MI, East HE, et al. The metabolic syndrome and 11-year risk of incident cardiovascular disease in the atherosclerosis risk in communities study. Diabetes Care 2005;28:385-90.
15. Iglseder B, Cip P, Malaimare L, Ladurner G, Paulweber B. The metabolic syndrome is a stronger risk factor for early carotid atherosclerosis in women than in men. Stroke 2005;36:1212-7.
16. Namekata T, Suzuki K, Ishizuka N, Shirai K. Establishing baseline criteria of cardio-ankle vascular index as a new indicator of arteriosclerosis: a cross-sectional study. BMC Cardiovasc Disord 2011;11:51.
17. Lee SY, Park HS, Kim DJ, Han JH, Kim SM, Cho GJ, et al.
Appropriate waist circumference cutoff points for central obesity in Korean adults. Diabetes Res Clin Pract 2007;75:72-80.
18. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412-9.
19. Hu H, Cui H, Han W, Ye L, Qiu W, Yang H, et al. A cutoff point for arterial stiffness using the cardio-ankle vascular index based on carotid arteriosclerosis. Hypertens Res 2013;36:334-41.
20. Shirai K, Hiruta N, Song M, Kurosu T, Suzuki J, Tomaru T, et al.
Cardio-ankle vascular index(CAVI) as a novel indicator of arterial stiffness: theory, evidence and perspectives. J Atheroscler Thromb 2011;18:924-38.
21. O’Rourke M. Arterial stiffness, systolic blood pressure, and logical
treatment of arterial hypertension. Hypertension 1990;15:339-47.
22. Mendelsohn ME. Protective effects of estrogen on the cardiovascular system. Am J Cardiol 2002;89:12E-7E; discussion 7E-8E.
23. Peiris AN, Aiman EJ, Drucker WD, Kissebah AH. The relative contributions of hepatic and peripheral tissues to insulin resistance in hyperandrogenic women. J Clin Endocrinol Metab 1989;68:715- 20.
24. Waddell TK, Dart AM, Gatzka CD, Cameron JD, Kingwell BA.
Women exhibit a greater age-related increase in proximal aortic stiffness than men. J Hypertens 2001;19:2205-12.
25. Kloting I, Kovacs P, van den Brandt J. Sex-specific and sex- independent quantitative trait loci for facets of the metabolic
syndrome in WOKW rats. Biochem Biophys Res Commun 2001;284:150-6.
26. Seda O, Tremblay J, Gaudet D, Brunelle PL, Gurau A, Merlo E, et al. Systematic, genome-wide, sex-specific linkage of cardiovascular traits in French Canadians. Hypertension 2008;51:1156-62.
27. Mattace-Raso FU, van der Cammen TJ, Hofman A, van Popele NM, Bos ML, Schalekamp MA, et al. Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam Study. Circulation 2006;113:657-63.
28. Kadota K, Takamura N, Aoyagi K, Yamasaki H, Usa T, Nakazato M, et al. Availability of cardio-ankle vascular index(CAVI) as a screening tool for atherosclerosis. Circ J 2008;72:304-8.