19 Received: March 8, 2010, Accepted for Publication: March 18, 2010
*Corresponding author: In-Jae Oh, 519-809, Department of Pulmonary and Critical Care Medicine, Lung and Esophageal Cancer Clinic, Chonnam National University Hwasun Hospital, Phone: +82-61-379-7617, FAX: +82-61-379- 7619, E-mail: [email protected]
Chonnam Medical Journal Vol. 46, No. 1, pp. 19∼24 DOI: 10.4068/cmj.2010.46.1.19
Risk Factors for Fatal Hemoptysis after Concurrent Chemoradiation Therapy in Patients with Non-Small Cell Lung Carcinoma
Yoon-Hee Kim1, Eun-Young Kim1, Hee-Jung Ban1,2, In-Jae Oh1,2*, Kyu-Sik Kim1,2, Young-Chul Kim1,2 and Sung-Ja Ahn2,3
1Department of Internal Medicine, Chonnam National University Medical School, Gwangju,
2Lung and Esophageal Cancer Clinic, Chonnam National University Hwasun Hospital, Jeonnam,
3Department of Radiation Oncology, Chonnam National University Medical School, Gwangju, Korea
Massive hemoptysis is a serious complication leading to death in lung cancer patients. To investigate whether fatal hemoptysis is associated with concurrent chemoradiation therapy (CCRT), the authors retrospectively analyzed risk factors for fatal hemoptysis after CCRT. The records of 150 patients (129 men and 21 women) with non-small cell lung cancer (NSCLC) who were treated with CCRT from April 2004 to June 2007 were reviewed for toxicity- related fatal hemoptysis. The dose of radiation ranged from 2500 to 9660 cGy, with a mean dose of 6506 cGy. Paclitaxel (45 mg/m2), docetaxel (20 mg/m2), or gemcitabine (350 mg/m2) with cisplatin 20 mg/m2 given weekly for 6 weeks during radiation therapy. Out of 150 patients, 12 (8.0%) patients died of fatal hemoptysis, and all but three had central and squamous tumors. Most of them had radiation pneumonitis or fibrosis. Median time to fatal hemoptysis after CCRT was 9.6 months, ranging from 84 days to 22 months. From a univariate analysis, the significant clinical factors for fatal hemoptysis were central location (p=0.004), central & squamous tumor (p=0.004), poor performance status (p=0.018), and chemotherapy with gemcitabine (p=0.017). From a multivariate analysis, poor performance status (OR=6.698, 95% CI, 1.083∼41.430, p=0.041) and central location (OR=6.976, 95% CI, 1.414∼
34.430, p=0.017) were independent risk factors. Centrally located tumors and poor performance status have been found to be significantly associated with fatal hemoptysis in NSCLC patients treated with CCRT.
Key Words: Hemoptysis; Lung neoplasms; Drug therapy; Radiotherapy
Introduction
Chemoradiation therapy has been the mainstay of treatment in patients with locally advanced non-small cell lung carcinoma (NSCLC), due to its superiority over radiation therapy alone.1-4 Chemoradiation therapy can be
administered patients sequentially or concurrently. A number of studies have found that concurrent chemoradiation therapy (CCRT) appears to outweigh sequential therapy in view of treatment effectiveness.5,6 It has been shown that CCRT can be considered tolerable under the maximum tolerated dose.7
We have defined fatal hemoptysis as a massive he- moptysis resulting in death of the patient. No study has been conducted to investigate the risk factors associated with fatal hemoptysis following CCRT in locally advanced
NSCLC patients.
From the unpredictable perspective of fatal hemoptysis, we decided to perform a retrospective investigation. We observed 12 cases of fatal hemoptysis during a 3-year period in our hospital, which led to death after CCRT in patients with locally advanced NSCLC. We analyzed risk factors associated with fatal hemoptysis after CCRT in NSCLC patients.
Materials and Methods
A retrospective review of patients diagnosed as having NSCLC and treated with CCRT was carried out at a single referral hospital. The records of 150 patients (129 men and 21 women) with locally advanced NSCLC who were treated with CCRT between April 2004 and June 2007 in Chonnam National University Hospital, Hwasun were investigated. Three-dimensional conformal radiation therapy techniques were used on all patients in accordance with recommended practice by the National Comprehensive Cancer Network (NCCN) guidelines.8,9 Radiation doses ranged from 2500 to 9660 cGy, with a mean dose of 6506 cGy. Paclitaxel (45 mg/m2), docetaxel (20 mg/m2), or gem- citabine (350 mg/m2) with cisplatin 20 mg/m2 were ad- ministered weekly, combined with radiation therapy for 6 weeks. Those chemotherapeutic agents were assigned with consideration for clinical settings with informed con- sents from patients. The dose volume histogram for organs included the lungs, esophagus, heart, and spinal cord were evaluated in order to minimize normal tissue toxicity.10-14 We tried to analyze the clinical factors related to fatal hemoptysis, such as histologic findings, involvement of central airways, response to treatments, performance status, pulmonary function, chemotherapeutic agents, and radiation doses.
We described the airway involvement as central and peripheral. When the tumor involved was proximal to segmental bronchus, the main bronchus or lobar bronchus was classified as central airway involvement. Bronchoscopic findings and computerized tomography (CT) scans were available for assessment of airway involvement. Some of
the patients underwent surgery following a down-staging from CCRT so we included surgery in the clinical para- meters. Regarding response to treatment, we tried to find out whether or not the tumor progressed during the final CT scans. Performance status could be characterized using the Eastern cooperative oncology group (ECOG) perfor- mance scale.15 We defined poor performance status by more than 2 points on the ECOG performance scale. Treatment could be initiated in patients with poor performance status after providing them with the information on the risks and benefits of CCRT. Regarding air-flow limitation, we classified patients as having chronic obstructive pulmonary disease (COPD) or not using the global initiative for chronic obstructive lung disease (GOLD) guidelines.16 Following completion of a 6-week course of CCRT, consolidation chemotherapy was given according to the response to treatment under the informed consent of patients while providing the information on the risks and advantages of the treatment.
Groups were classified as either fatal hemoptysis or non fatal hemoptysis. We implemented a univariate analysis from Pearson’s Chi-square, Fisher’s exact tests for categorical data including histology, location, tumor progression, gender, presence of air flow limitation, performance status, difference among chemotherapeutics, and consolidation chemotherapy, and by unpaired Student’s t-test for continuous data, including age, smoking in pack-years, and radiation doses.
We conducted a multivariate analysis with a logistic regression following an adjustment for clinical factors. A p value
<0.05 was assumed to be statistically significant.
Results
Out of the 150 patients, 12 (8.0%) died of fatal hemoptysis.
There were 10 men and 2 women, and their median age at diagnosis was 62 years. All except 3 patients had central and squamous tumors. The major symptoms prior to fatal hemoptysis were infection-related fever (n=6), dyspnea (n=5), and chest pain (n=1). All patients had radiation pneumonitis and/or radiation fibrosis. A Bronchoscopy performed prior to fatal hemoptysis demonstrated radiation
Table 1. Clinical characteristics of the patients that presented with fatal hemoptysis
Case Age
(years) Gender Histology Stage Location Response PS COPD CTX/con RT dose Interval
(days) Symptom RP Survival (days)
1 59 F ADC IV Central SD 0 No GP/+ 4,500 305 Infection + 365
2 64 M SQC IIIB Central PD 0 Yes GP/+ 5,280 358 Dyspnea + 421
3 70 M NSCLC IIIA Central PD 1 Yes TP/+ 4,800 84 Infection + 1897
4 69 M SQC IIIB Central SD 1 Yes TP/+ 8,400 189 Dyspnea + 251
5 66 M SQC IIIB Central PD 2 Yes TP/+ 6,600 203 Infection + 254
6 62 M SQC IIIA Peripheral PD 1 Yes TP/- 6,000 659 Dyspnea + 755
7 53 F SQC IIIB Central PD 1 Yes DP/+ 7,320 605 Dyspnea + 676
8 66 M SQC IIIB Central PR 1 Yes TP/+ 7,700 197 Infection + 275
9 59 M SQC IIIB Central SD 1 Yes GP/+ 8,100 272 Infection + 345
10 54 M SQC IIIB Central PD 1 Yes GP/+ 7,800 210 Chest pain + 281
11 61 M SQC IIIB Central SD 2 Yes TP/+ 7,700 220 Dyspnea + 254
12 61 M SQC IIIB Central SD 2 Yes GP/+ 7,700 149 Infection + 182
PS, performance status; CTX/con, chemotherapy/consolidation; RT dose, dose of radiation therapy; Interval, the interval between fatal hemoptysis and completion of CCRT; Symptom, the last symptom before fatal hemoptysis; Survival, the survival period after pathologic confirmation; F, female; M, male; SQC, squamous cell carcinoma; ADC, adenocarcinoma; NSCLC, non-small cell lung carcinoma; PR, partial response; SD, stable disease; PD, progressive disease; GP, gemcitabine+cisplatin; TP, paclitaxel+cisplatin; DP, docetaxel+cisplatin; RP, radiation pneumonitis.
Fig. 1. Sixty two-year old man with locally advanced NSCLC treated with concurrent chemoradiation therapy. (A) Chest CT scan with lung parenchymal window shows ground glass opacities associated with consolidation representing radiation pneumonitis. (B) Bronchoscopy performed prior to fatal hemoptysis in a patient who died of fatal toxicity demonstrats whitish necrotic material covering bronchial mucosa indicative of radiation induced bronchial injury.
induced injury in 3 cases. Representative CT images demonstrating radiation pneumonitis and bronchoscopic findings depicting radiation-induced bronchial injury are presented in Fig. 1. Median time to fatal hemoptysis after CCRT was 9.5 months, ranging from 84 days to 22 months.
Most of them had a stage IIIB or IV disease, except for two stage-IIIA patients. For chemotherapeutic agents, Paclitaxel was given to 6 patients, Gemcitabine to 5 patients, and Docetaxel to 1 patient. Final CT scannings
revealed a progression state in 6 patients. Demographic characteristics of the patients are shown in Table 1.
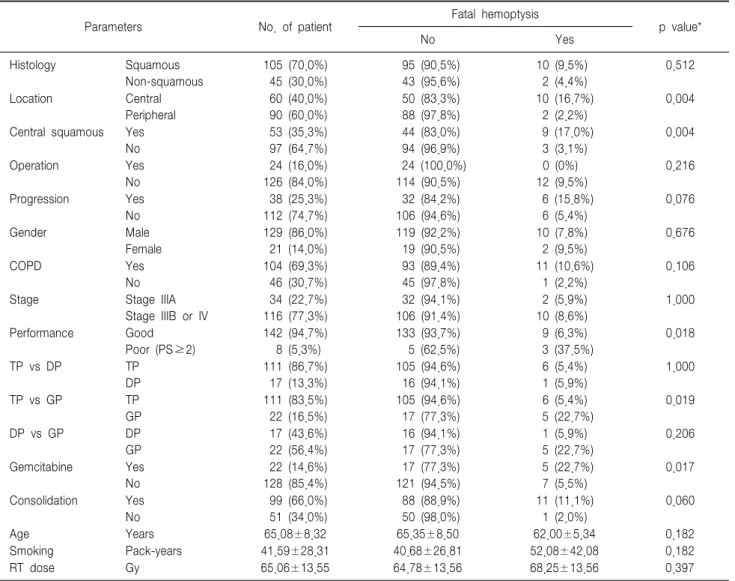
A univariate analysis demonstrated a statistically significant correlation between fatal hemoptysis and central location (p=0.004), central & squamous tumor (p=0.004), poor performance status (p=0.018), and chemotherapy with gemcitabine (p=0.017) (Table 2). The difference between squamous and non-squamous histology failed to find a significant association with fatal hemoptysis. However, the
Table 2. Incidence of fatal hemoptysis according to clinical parameters
Parameters No. of patient Fatal hemoptysis
p value*
No Yes
Histology Squamous
Non-squamous
105 (70.0%) 45 (30.0%)
95 (90.5%) 43 (95.6%)
10 (9.5%) 2 (4.4%)
0.512
Location Central
Peripheral
60 (40.0%) 90 (60.0%)
50 (83.3%) 88 (97.8%)
10 (16.7%) 2 (2.2%)
0.004
Central squamous Yes No
53 (35.3%) 97 (64.7%)
44 (83.0%) 94 (96.9%)
9 (17.0%) 3 (3.1%)
0.004
Operation Yes
No
24 (16.0%) 126 (84.0%)
24 (100.0%) 114 (90.5%)
0 (0%) 12 (9.5%)
0.216
Progression Yes
No
38 (25.3%) 112 (74.7%)
32 (84.2%) 106 (94.6%)
6 (15.8%) 6 (5.4%)
0.076
Gender Male
Female
129 (86.0%) 21 (14.0%)
119 (92.2%) 19 (90.5%)
10 (7.8%) 2 (9.5%)
0.676
COPD Yes
No
104 (69.3%) 46 (30.7%)
93 (89.4%) 45 (97.8%)
11 (10.6%) 1 (2.2%)
0.106
Stage Stage IIIA
Stage IIIB or IV
34 (22.7%) 116 (77.3%)
32 (94.1%) 106 (91.4%)
2 (5.9%) 10 (8.6%)
1.000
Performance Good
Poor (PS≥2)
142 (94.7%) 8 (5.3%)
133 (93.7%) 5 (62.5%)
9 (6.3%) 3 (37.5%)
0.018
TP vs DP TP
DP
111 (86.7%) 17 (13.3%)
105 (94.6%) 16 (94.1%)
6 (5.4%) 1 (5.9%)
1.000
TP vs GP TP
GP
111 (83.5%) 22 (16.5%)
105 (94.6%) 17 (77.3%)
6 (5.4%) 5 (22.7%)
0.019
DP vs GP DP
GP
17 (43.6%) 22 (56.4%)
16 (94.1%) 17 (77.3%)
1 (5.9%) 5 (22.7%)
0.206
Gemcitabine Yes
No
22 (14.6%) 128 (85.4%)
17 (77.3%) 121 (94.5%)
5 (22.7%) 7 (5.5%)
0.017
Consolidation Yes No
99 (66.0%) 51 (34.0%)
88 (88.9%) 50 (98.0%)
11 (11.1%) 1 (2.0%)
0.060
Age Years 65.08±8.32 65.35±8.50 62.00±5.34 0.182
Smoking Pack-years 41.59±28.31 40.68±26.81 52.08±42.08 0.182
RT dose Gy 65.06±13.55 64.78±13.56 68.25±13.56 0.397
*Univariate analysis by Pearson’s Chi-square and Fisher exact tests for categorical data expressed by frequency and percentage & by unpaired Student's t-test for continuous data expressed by mean and standard deviation.
Table 3. Multivariate analysis of clinical factors
Parameters aOR* 95% CI p value
Poor performance
Chemotherapy with gemcitabine Central location
6.698 3.953 6.976
1.083∼41.430 0.986∼15.843 1.414∼34.430
0.041 0.052 0.017 aOR, adjusted odds ratio; CI, confidence interval.
*Adjusted for chemotherapy with gemcitabine & central squamous by multivariate logistic regression.
univariate anlaysis demonstrated that whether or not a squamous tumor was centrally located, it was meaningfully pertinent to fatal hemoptysis. No significant correlations with fatal hemoptysis were observed in surgery, tumor progression, gender, age, smoking amount, radiation dose, pulmonary function, or stage discrimination. With regard to chemotherapy, whether or not consolidation chemotherapy was added, there was no association with fatal toxicity, while gemcitabine appeared to be likened to fatal toxicity by univariate analysis.
Based on the multivariate analysis, clinical factors with a significant correlation with fatal hemoptysis included poor performance status (OR=6.698, 95% CI, 1.083∼41.430,
p=0.041) and central location (OR=6.976, 95% CI, 1.414
∼34.430, p=0.017) (Table 3). Chemotherapy with gemci- tabine and squamous tumor histology were not found to be connected with fatal hemoptysis. We were able to demonstrate that a centrally located tumor and poor perfor-
mance status have been found to be significantly associated with fatal hemoptysis in locally advanced NSCLC patients treated with CCRT.
Discussion
A Phase I trial found that CCRT was tolerable under the maximal tolerated dose without reporting a fatal toxicity, such as fatal hemoptysis, which can lead to sudden death.7 In our retrospective review of CCRT in locally advanced NSCLC patients, clinical factors, including a centrally located tumor and poor performance status, were shown to be associated with fatal toxicity.
Mehta and Dweik17 reported the role of radiation in view of necrosis of the bronchus in their description of 4 cases of such necrosis that occurred as a consequence of radiation therapy, 2 of which were presented with massive hemoptysis. Patients had a pulmonary infection in the set up of squamous cell carcinoma, which was thought to be a marker of poor prognosis. Panos et al18 found the factors associated with fatal pulmonary hemorrhage were more likely to occur when hemoptysis results from a complication of fungal pneumonia and necrotic bronchogenic squamous cell carcinoma. In our report, histology does not appear to be linked to fatal hemoptysis, but central location seems to be a marker of poor prognosis regarding fatal hemoptysis.
In addition, pulmonary infection may be linked to fatal hemoptysis for 6 out of 12 patients with pulmonary infection prior to fatal hemoptysis, as presented in Table 1.
At present, no clinical trial has supported one specific agent over others in the setting of CCRT.6,19-22 In this study, gemcitabine had mixed results according to to both uni- variate and multivariate analyses. The relatively small number and uneven distribution of chemotherapeutics in patients with fatal hemoptysis might be a limitation of statistical power to support the result. We found only one case report about gemcitabine-associated diffuse alveolar hemorrhage in the literature.23 We looked for common radiologic findings in patients with fatal hemoptysis.
However, chest CT scan findings in patients who died of
fatal hemoptysis varied from cavitary changes and concentric bronchial wall thickening to various types of radiation injury, because three-dimensional conformal radiation therapy can result in an unusual distribution of findings.21 We think that further studies on radiologic findings, in addition to the correlation of chemotherapeutic agents with toxicity following CCRT, would be required.
Fatal hemoptysis after CCRT in advanced NSCLC patients can happen unexpectedly and culminate in a fatal consequence. Hence, when we consider CCRT in patients with centrally located NSCLC, especially when performance status is not good, we should take into account the possibility of fatal hemoptysis.
Acknowledgements
The authors are grateful to the multidisciplinary team members of Lung and Esophageal Cancer Clinic of Chonnam National University Hwasun Hospital for the comprehensive patient care.
References
1. Dillman RO, Seagren SL, Propert KJ, Guerra J, Eaton WL, Perry MC, et al. A randomized trial of induction chemotherapy plus high-dose radiation versus radiation alone in stage III non-small-cell lung cancer. N Engl J Med 1990;323:940-5.
2. Le Chevalier T, Arriagada R, Quoix E, Ruffie P, Martin M, Tarayre M, et al. Radiotherapy alone versus combined chemotherapy and radiotherapy in nonresectable non-small-cell lung cancer: first analysis of a randomized trial in 353 patients. J Natl Cancer Inst 1991;83:417-23.
3. Schaake-Koning C, van den Bogaert W, Dalesio O, Festen J, Hoogenhout J, van Houtte P, et al. Effects of concomitant cisplatin and radiotherapy on inoperable non-small-cell lung cancer. N Engl J Med 1992;326:524-30.
4. Dillman RO, Herndon J, Seagren SL, Eaton WL Jr, Green MR. Improved survival in stage III non-small-cell lung cancer: seven-year follow-up of cancer and leukemia group B (CALGB) 8433 trial. J Natl Cancer Inst 1996;88:1210-5.
5. Furuse K, Fukuoka M, Kawahara M, Nishikawa H, Takada Y, Kudoh S, et al. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with mitomycin, vindesine, and cisplatin in unresectable stage III non-small-cell lung cancer. J Clin Oncol 1999;17:2692-9.
6. Socinski MA, Rosenman JG, Halle J, Schell MJ, Lin Y, Russo S, et al. Dose-escalating conformal thoracic radiation therapy with induction
and concurrent carboplatin/paclitaxel in unresectable stage IIIA/B nonsmall cell lung carcinoma: a modified phase I/II trial. Cancer 2001;92:1213-23.
7. Schild SE, McGinnis WL, Graham D, Hillman S, Fitch TR, Northfelt D, et al. Results of a Phase I trial of concurrent chemotherapy and escalating doses of radiation for unresectable non-small-cell lung cancer.
Int J Radiat Oncol Biol Phys 2006;65:1106-11.
8. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Non-Small Cell Lung Cancer V.2.2010. available from http://www.nccn.org/professionals/physician_gls/PDF/nscl.pdf. accessed Mar 3, 2010.
9. Emami B. Three-Dimensional Conformal Radiation Therapy In Bronchogenic Carcinoma. Semin Radiat Oncol 1996;6:92-7.
10. Graham MV, Purdy JA, Emami B, Harms W, Bosch W, Lockett MA, et al. Clinical dose-volume histogram analysis for pneumonitis after 3D treatment for non-small cell lung cancer (NSCLC). Int J Radiat Oncol Biol Phys 1999;45:323-9.
11. Kong FM, Hayman JA, Griffith KA, Kalemkerian GP, Arenberg D, Lyons S, et al. Final toxicity results of a radiation-dose escalation study in patients with non-small-cell lung cancer (NSCLC): predictors for radiation pneumonitis and fibrosis. Int J Radiat Oncol Biol Phys 2006;65:1075-86.
12. Wang S, Liao Z, Wei X, Liu HH, Tucker SL, Hu CS et al. Analysis of clinical and dosimetric factors associated with treatment-related pneumonitis (TRP) in patients with non-small-cell lung cancer (NSCLC) treated with concurrent chemotherapy and three-dimensional conformal radiotherapy (3D-CRT). Int J Radiat Oncol Biol Phys 2006;66:1399-407.
13. Hernando ML, Marks LB, Bentel GC, Zhou SM, Hollis D, Das SK, et al. Radiation-induced pulmonary toxicity: a dose-volume histogram analysis in 201 patients with lung cancer. Int J Radiat Oncol Biol Phys 2001;51:650-9.
14. Kim TH, Cho KH, Pyo HR, Lee JS, Zo JI, Lee DH, et al. Dose-volumetric parameters for predicting severe radiation pneumonitis after three- dimensional conformal radiation therapy for lung cancer. Radiology 2005;
235:208-15.
15. Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982;5:649-55.
16. Pauwels RA, Buist AS, Calverley PM, Jenkins CR, Hurd SS;GOLD Scientific Committee. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am J Respir Crit Care Med 2001;163:1256-76.
17. Mehta AC, Dweik RA. Necrosis of the bronchus. Role of radiation.
Chest 1995;108:1462-6.
18. Panos RJ, Barr LF, Walsh TJ, Silverman HJ. Factors associated with fatal hemoptysis in cancer patients. Chest 1988;94:1008-13.
19. Belani CP, Choy H, Bonomi P, Scott C, Travis P, Haluschak J, et al. Combined chemoradiotherapy regimens of paclitaxel and carboplatin for locally advanced non-small-cell lung cancer: a randomized phase II locally advanced multi-modality protocol. J Clin Oncol 2005;23:5883-91.
20. Albain KS, Crowley JJ, Turrisi AT 3rd, Gandara DR, Farrar WB, Clark JI, et al. Concurrent cisplatin, etoposide, and chest radiotherapy in pathologic stage IIIB non-small-cell lung cancer: a Southwest Oncology Group phase II study, SWOG 9019. J Clin Oncol 2002;20:3454-60.
21. Vokes EE, Herndon JE 2nd, Crawford J, Leopold KA, Perry MC, Miller AA, et al. Randomized phase II study of cisplatin with gemcitabine or paclitaxel or vinorelbine as induction chemotherapy followed by concomitant chemoradiotherapy for stage IIIB non-small-cell lung cancer:
cancer and leukemia group B study 9431. J Clin Oncol 2002;20:4191-8.
22. Choi YW, Munden RF, Erasmus JJ, Park KJ, Chung WK, Jeon SC, et al. Effects of radiation therapy on the lung: radiologic appearances and differential diagnosis. Radiographics 2004;24:985-97.
23. Carron PL, Cousin L, Caps T, Belle E, Pernet D, Neidhardt A, et al.
Gemcitabine-associated diffuse alveolar hemorrhage. Intensive Care Med 2001;27:1554.