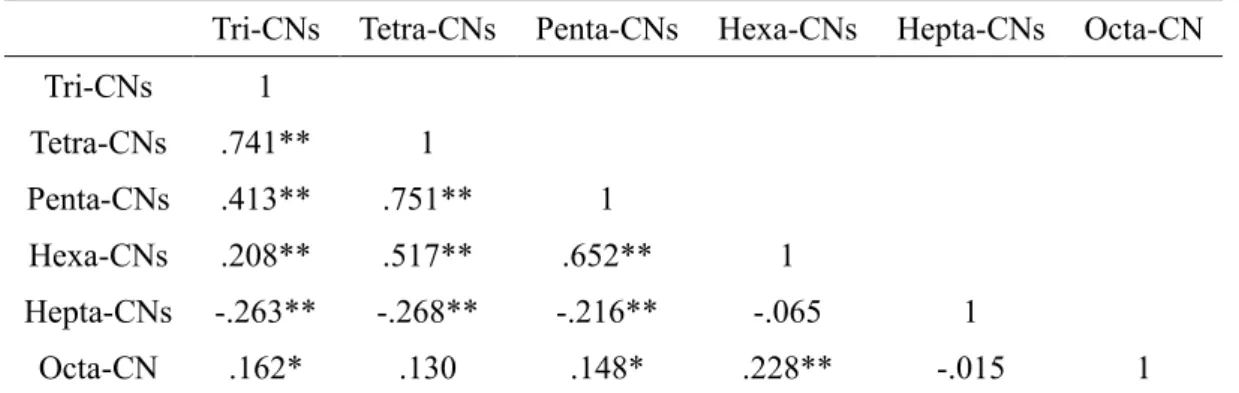
There have been a few studies of PCNs in foods; however, most of them have focused on fishery products, and only a single study has been done in Korea. From the results, contamination levels and correlation between PCNs in food were investigated and dietary intake from food was estimated. In addition, the total combined dietary intake of PCNs, PCDD/Fs, and DL-PCBs among the Korean population was estimated using previous results.
The concentration levels of PCNs in food groups in this study are similar to those in other countries. The relative composition of hexa-CNs was the highest for Σ TEQPCN in foods due to their high relative strength.
INTRODUCTION
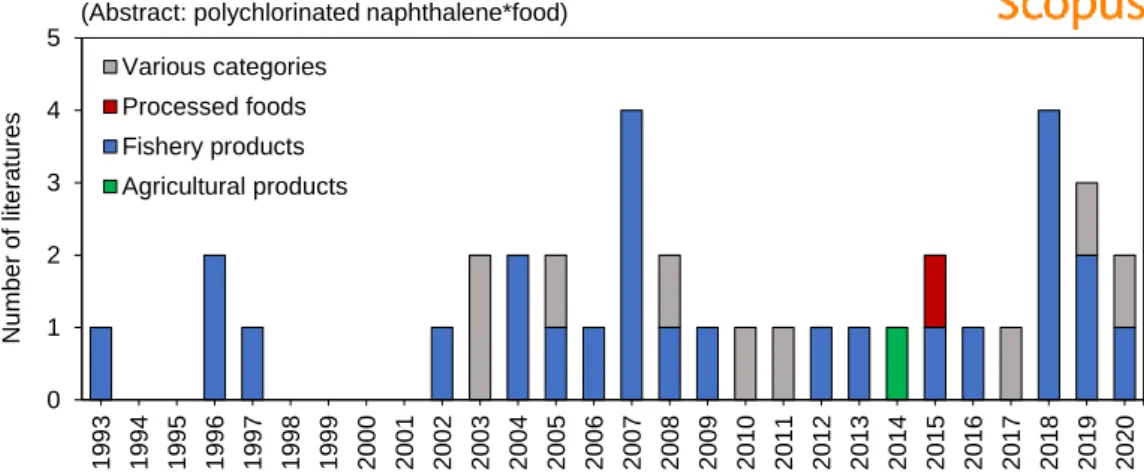
Therefore, it is important to estimate the dietary intake of PCNs for PCN exposure assessment. Number of literature on the analysis of PCNs in food samples (the written literature on concentration levels and intake in food were selected; review papers were included). There have been several studies monitoring PCN levels in food since 1993 (Figure 4).
Only one study in Korea has examined the quantitative levels of PCNs in food and evaluated dietary intake of PCNs (Kim et al., 2018), but this study was only conducted on fishery products. Therefore, it would be necessary to monitor the levels of PCNs and estimate the intake of PCNs in different food groups, including agricultural products and processed foods.
MATERIALS AND METHOD
- Sampling
- Sample selection
- Sample collection
- Analytical procedure
- Quality assurance and quality control (QA/QC)
- Statistical analysis
- Dietary intake assessment
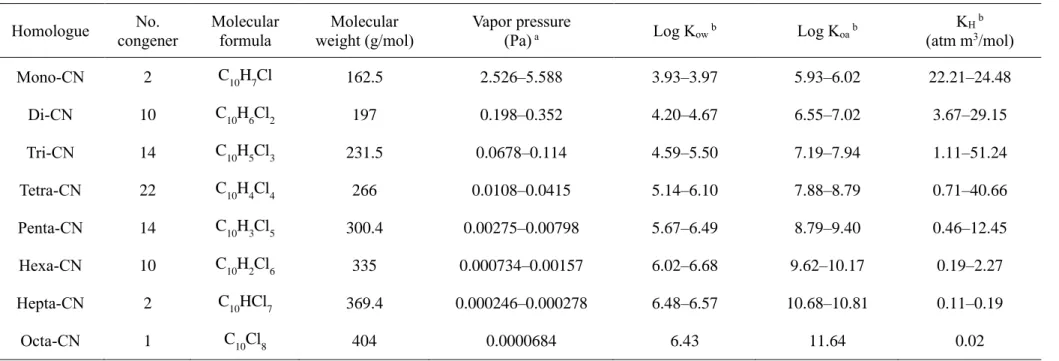
The extracts were exchanged into n-hexane and a gravimetric method was used to measure the lipid content of each sample. The extracts were then concentrated to 10 mL using a rotary evaporator (Hei-VAP Advantage, Heidolph, Germany) and mixed with 20 mL of sulfuric acid (96% purity) to remove the interrupting compounds for analysis. After repeated sulfuric acid treatments, the extracts were neutralized with 100 mL of ultrapure water until the pH reached 6–7.
After the extracts were eluted with n-hexane (200 mL), the eluents were concentrated to 2 mL using a rotary evaporator. The extracts were concentrated to approx. 25 μL using a nitrogen vaporizer (MGS-2200, Eyela, Japan), and then a 13C12-labeled internal standard (ED-910, 1000 pg, Cambridge Isotope Laboratories Inc.) was added before instrumental analysis.
RESULTS AND DISCUSSION
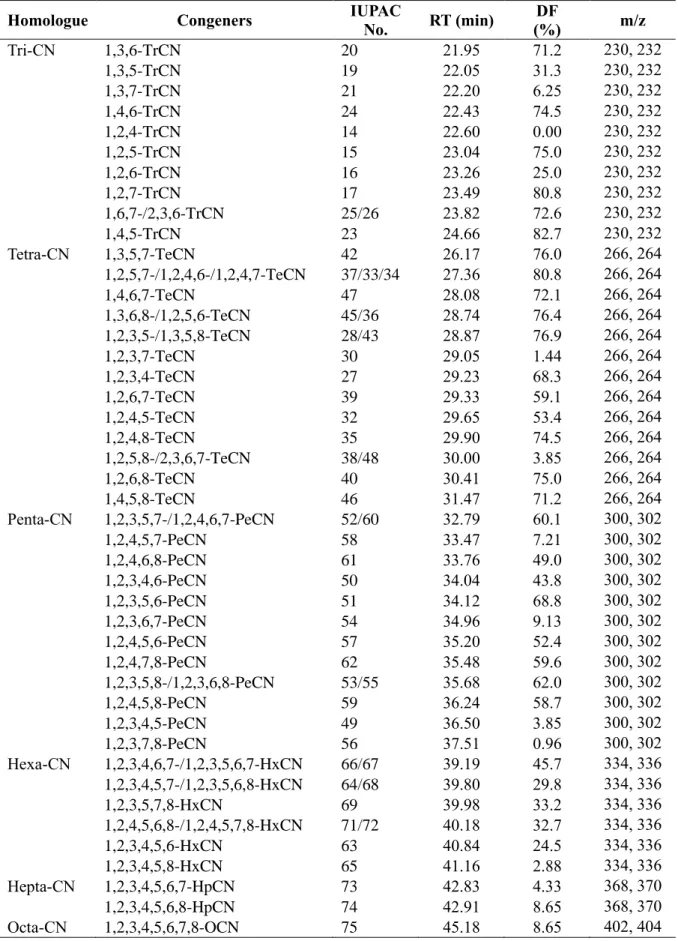
Levels of 55 PCNs in food
- Agricultural products
- Fishery products
- Processed foods
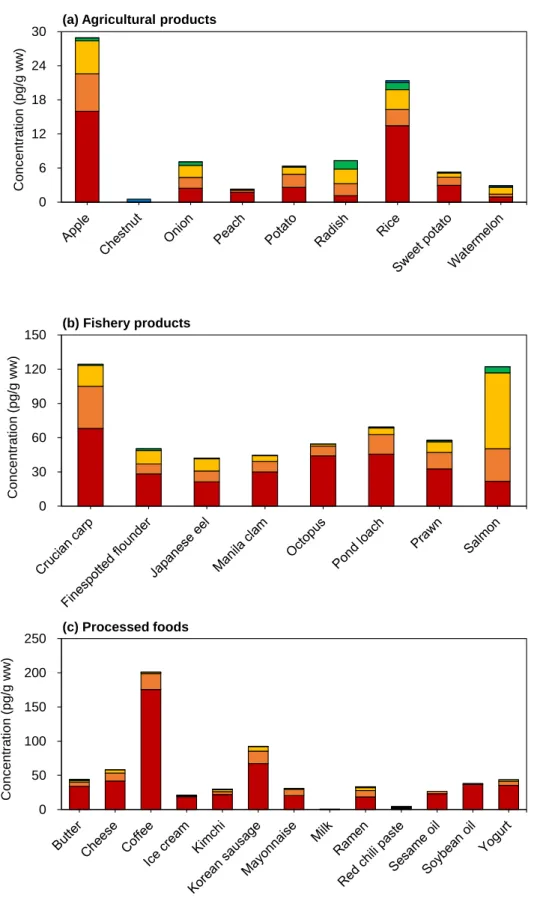
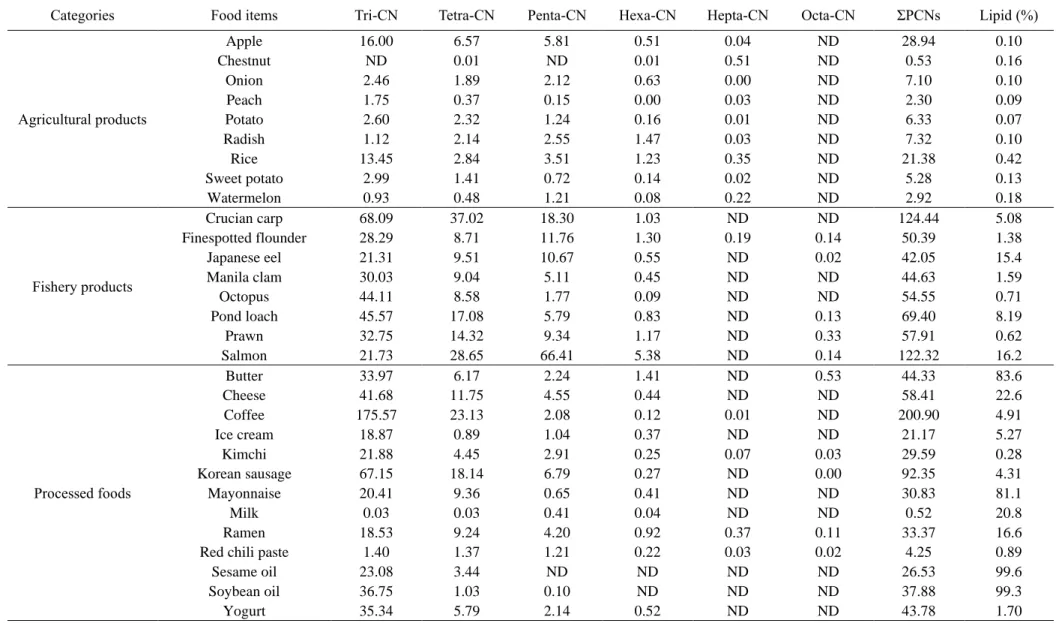
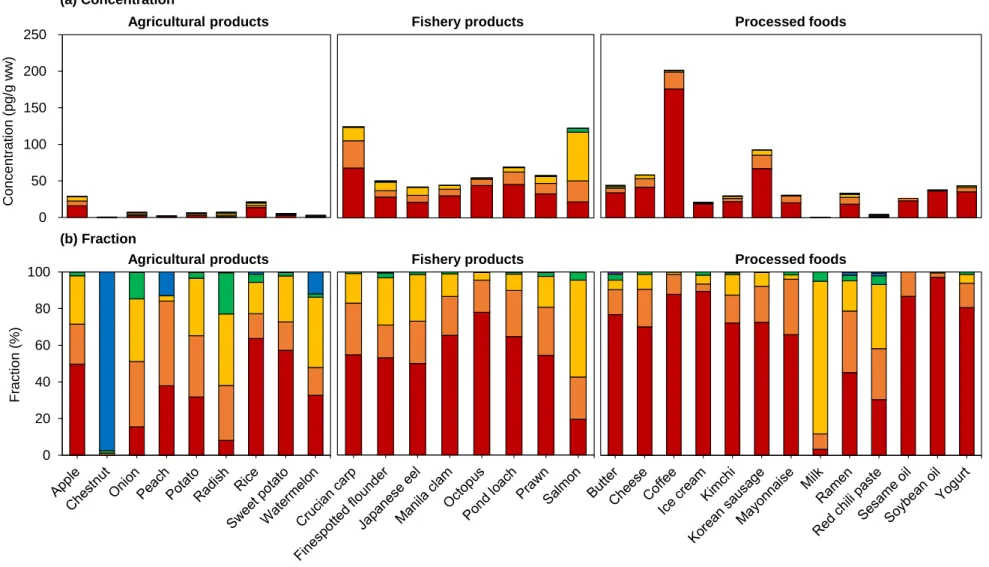
Concentrations of the total Σ55 PCNs in food samples from (a) agricultural products, (b) fishery products, and (c) processed foods (n=7; n=6 for cheese and shrimp). a) Agricultural products (b) Fishery products (c) Processed food. According to the previous studies, dioxin uptake by plants is from the atmospheric gas phase rather than absorption from the soil through the roots (Uegaki et al., 2006; Wagrowski & Hites, 1998). Among them are the edible parts of watermelon, chestnut, and peach covered with rind of watermelon, cusule (called 'burr') of chestnut, and indumentum of peach (Lu et al., 2015).
In addition, the lenses on the apple peel serve as a site of gas exchange with air (Khanal et al., 2020; Veraverbeke et al., 2003), which could increase PCN exposure. Meanwhile, Pakistani studies for tri-CN to octa-CN reported 24.6 pg/g ww in rice, which was higher than the present study (Mahmood et al., 2014). As the water-to-fish pathway via the gills is one of the main accumulation pathways for the bioaccumulation of POPs in fish (Burreau et al., 2004), poor water quality could influence the high accumulation of PCNs in the crucian.
Furthermore, a previous study reported that higher levels of POPs, including PCNs, were observed in some freshwater fish than in marine fish (Rose et al., 2015). Furthermore, a previous study reported that higher levels of PCNs and PCBs were observed in farmed salmon rather than wild salmon (Easton et al., 2002; Fernandes et al., 2010; Hites et al., 2004). Most of the salmon consumed in Korea is farmed in Norway, and this could have caused the high PCN levels in salmon in this study.
Crucian carp was relatively rich in CN-42 compared to other species, and this is similar to previous results (Gewurtz et al., 2018). Furthermore, in a previous study of PAHs in coffee beans (Houessou et al., 2007), higher concentrations of PAHs corresponded to roasted coffee beans rather than green beans, and higher levels of PAHs were observed at higher roasting temperatures. Concentrations of the PCNs in food samples of (a) agricultural products, (b) fishery products, and (c) processed foods.
Profiles of PCNs in food
- Homologue profiles
- Correlation among PCNs, PCDD/Fs and DL-PCBs
- Principal component analysis (PCA)
The distribution of PCNs in this study is similar to previous studies on rice and wheat in Pakistan (Mahmood et al., 2014) and fisheries in Spain (Llobet et al., 2007) and China (Cui et al., 2018). ), which concluded that tetra-CN and/or penta-CN were the predominant homologues from tetra-CN to octa-CN. In a study on fishery products with tri-CN to octa-CN, tri-CN and penta-CN were the dominant congeners (Jiang et al., 2007). Previous studies on different environmental matrices, including air (Xue et al., 2016), soil (Mahmood et al., 2014), and sediment (Lundgren et al., 2002), also found similar distribution patterns.
In a few studies of human related samples, tetra-CN and penta-CN are the major contributors in human serum (Park et al., 2010), while it was penta-CN and hexa-CN in breast milk (Pratt et al., 2013) and adipose tissue (Schiavone et al., 2010) with the highest contributions from CN-66/67. Most of the agricultural products, milk and red chili paste showed slightly different homologue profiles than the other products. Salmon is one of the higher trophic level organisms, while crucian carp belong to bottom feeders.
Similar results with high contributions of penta-CN and hexa-CN in higher trophic level organisms were observed in several previous studies (Gewurtz et al., 2018; Hanari et al., 2004; Helm et al., 2008). a) Mean concentrations and (b) proportions of PCNs in food samples in agricultural products, fishery products and processed food. As discussed above, CN-73, 74 and 75 (hepta-CN and octa-CN) bioaccumulate less because they are susceptible to photodegradation due to steric interactions of substituted chlorines in the 1,8- and 4,5-positions (Gewurtz et. et al., 2009; Helm et al., 2008). A previous study also did not observe a correlation between PCN and OCDD (Kim et al., 2018).
In the loading plot, principal component 1 (PC 1), explaining 36% of the total variance, has positive PC loading values for tetra- to octa-CN and negative values for tri-CNs. In the score plot, most of the agricultural products have positive PC 1 scores, indicating that agricultural products were much more correlated with tetra- to hexa-CNs. Meanwhile, negative PC 1 loading values were related to most of the processed foods, excluding milk, red chili paste, and ramen.
TEQ
Dietary intake
- Dietary intake of PCNs
- Total combined intake with PCDD/Fs and DL-PCBs
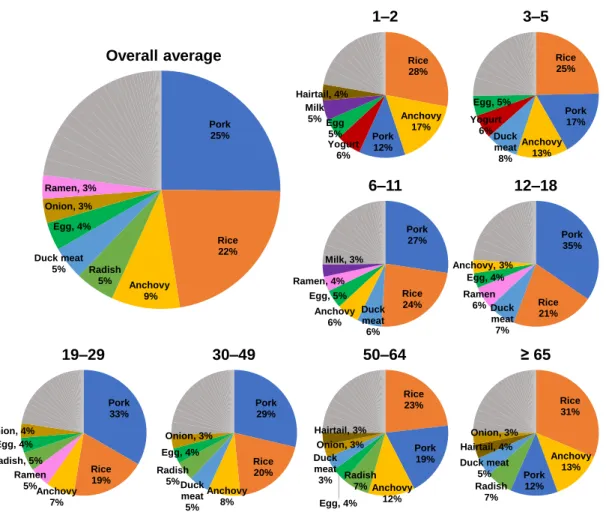
Distribution of dietary intake (pg-TEQ/kg body weight/day) of Σ11 TEQPCN in selected food samples. The sum of Σ11 TEQPCN intakes from these agricultural products ranged from 0.157 to 0.299 pg-TEQ/day. For the overall mean age, the highest intake of Σ11 TEQPCN from agricultural products corresponded to rice (63%), followed by radish (15%) and onion (9%), resulting from the high consumption of rice and onion and the high TEQ of radish.
Hairtail and mackerel also accounted for high intakes of Σ11 TEQPCN due to the high TEQ of hairtail and high consumption of mackerel. The contributions of seven livestock products to the intake of Σ11 TEQPCN are given in Figure 15. According to the overall average group, the highest contribution corresponded to pork (75%), followed by duck meat (14%), and eggs (11%).
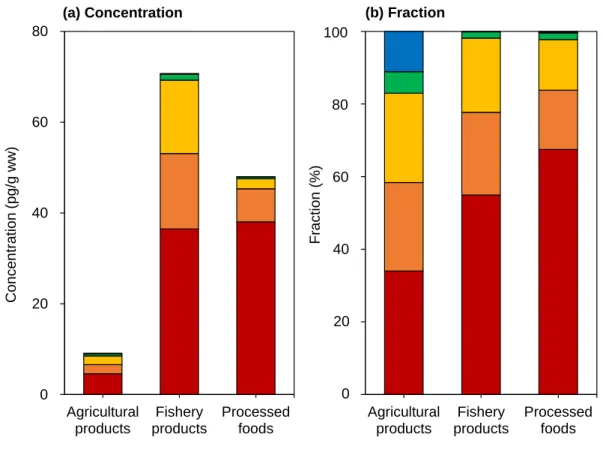
Based on total average consumption, agricultural products contributed the most to total Σ11 TEQPCN intake (36%), followed by livestock products (34%), fishery products (20%) and processed foods (10%). In contrast, in previous studies, fishery products usually contributed the most to total PCN intake (Fernandes et al., 2011; Martí-Cid et al., 2008; Zacs et al., 2020). However, in addition to PCN intake, other dioxin-like contaminants present in food must also be considered.
Therefore, the total combined intake of TEQ values from PCNs, PCDD/Fs and DL-PCBs was calculated and compared to the TWIs in 3.4.2. Mackerel (0.0541 pg-TEQ/kg bw/day) contributed the most to the combined dietary intake with a contribution of 63% of DL-PCBs. The total intake of most foods was highly influenced by DL-PCBs and PCDFs, whereas most of the agricultural products, excluding peaches, provided the highest contribution of PCNs to the overall total intake.
The sum of total combined intake in this study had a smaller contribution from DL-PCBs and larger contributions from PCDD/Fs and PCNs compared to the previous results. The ratio of ΣTEQPCN intake to ΣTEQPCDD/Fs intake in this study was an order of magnitude higher than that in previous studies.
CONCLUSION
Relative potencies of individual polychlorinated naphthalenes and halowax mixtures to induce Ah receptor-mediated responses. Polychlorinated naphthalenes: congener-specific analysis and source identification in a dated sediment core from Lake Thun, Switzerland. Concentrations and trophic enhancement of polychlorinated naphthalenes (PCNs) in marine fish from coastal Bohai, China.
A retrospective study of occurrence and human exposure to polychlorinated naphthalenes (PCNs), dibenzo-p-dioxins and furans (PCDD/Fs) and PCBs through cod liver products. Spatial distribution and bioaccumulation of polychlorinated naphthalenes (PCNs) in mussels and fish from the Gulf of Gdańsk, Baltic Sea. Polychlorinated naphthalenes (PCNs): congener-specific analysis, occurrence in food and dietary exposure in the UK.
Factors affecting trends in polychlorinated naphthalenes and other dioxin-like compounds in lake trout (Salvelinus namaycush) from Lake Ontario, North America. Occurrence and biomagnification of polychlorinated naphthalenes and non- and mono-ortho PCBs in sediments and organisms of Lake Ontario. Health risk assessment of polychlorinated biphenyls, polychlorinated dibenzo-p-dioxins and dibenzofurans, and polychlorinated naphthalenes in seafood from Guangzhou and Zhoushan, China.
Isomer-Specific Analysis and Toxic Assessment of Polychlorinated Naphthalenes in Soil, Sediment, and Biota Collected Near the Site of a Former Chlor-Alkali Plant. Human exposure to polychlorinated naphthalenes and polychlorinated diphenyl ethers from food in Catalonia, Spain: temporal trend. The validity of using a relative potency factor approach for risk management of dioxin-like polychlorinated naphthalenes.
Relative potencies of individual polychlorinated naphthalenes to induce dioxin-like responses in fish and mammals in vitro bioassays. Polychloronaphthalenes (PCNs) in food products in Latvia: Congener-specific analysis, occurrence and dietary exposure of the general population.