P1-45 / S. Y. Kim
• IMID 2009 DIGEST
Abstract
Photosensitive polyimide containing methoxy cinnamate derivatives as a photo-alignment layer is investigated. The anisotropy of alignment layer was induced by irradiation linearly polarized UV (LPUV). We studied the effect of the methoxy cinnamate groups on alignment LCs.
1.
Introduction
Uniform alignment of liquid crystals (LCs) is required for a successful operation of liquid crystal displays (LCDs). The interaction between polymer surfaces and liquid crystals (LCs) determines the alignment properties of a liquid crystal at its interface with a polymer layer. Polyimide is used as alignment layers because of their advantageous properties, such as excellent optical transparency, heat resistance and dimensional stability. Rubbed such PI film surfaces have been widely used for aligning LC molecules.
However, the rubbing method has some problems, such as the generation of electrostatic charges and dust formation. Rubbing free method for LC alignment, photo-alignment is expected to achieve the high resolution LCDs.
Photo-reactive polymers were actively studied for stability of LCDs alignment. The most prominent materials of these are derivatives of cinnamate groups. Later if was found that polyimide films exposed to linearly polarized UV (LPUV) irradiation give homogenous LC alignment perpendicular to the polarization direction of the light. In this study, the effect of methoxy cinnamate derivatives on LCs alignment layer is studied.
2. Experimental
4-Methoxy cinnamic acid, 2-methoxy cinnamic acid
and 2,5-dimethoxy cinnamic acid were irradiated and the absorption spectra were obtained with irradiation time in solubilized in chloroform.
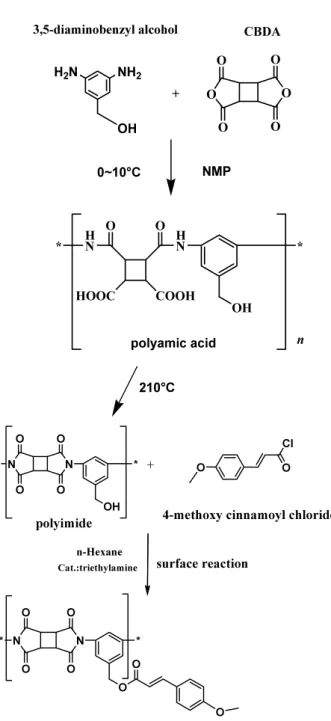
Polymerization processes are shown in Figure 1. 3,5-Diaminobenzly alcohol was polymerized with 1,2,3,4-cycolbutanetetrcatboxylic dianhydride (CBDA) using N-metyl-2-pyrrolidinon (NMP) as a solvent at 0℃
-10℃ for 12-24hr to obtain the polyamic acid. The
polyamic acid was imidized at 210℃ for 1hr.
The polyamic acid was spin coated onto indium-tin-oxide (ITO) coated glass substrate with spin coater. The polyamic acid films were pre-baked on the temperature-controlled hot plate at 90℃ for 30min.
The polyamic acid film substrates were hard-baked in the oven at 210℃ for 1hr to convert to the polyimide.
A mixture of 4-methoxy cinnamic acid and thionyl chloride was stirred under reflux until the disappearance of the starting materials as evidenced by TLC about 4hr. After reaction, the excess thionyl chloride was removed in vacuum. 2-methoxy cinnamic acid and 2,5-dimethoxy cinnamic acid were prepared the above method. The polyimide film reacted with 4-methoxy cinnamoyl chloride in dichloride methane (MC) using triethylamin as a catalyst by interfacial reaction.
Linearly polarized ultraviolet light (LPUV) was irradiated for 30min with 200W super pressure short arc mercury lamp equipped polarizer (LUMATEC medel SUV-DC-P, Glan-Taylor polarizer).
Parallel LC cells were fabricated with the alignment layers with the cell gaps of approximately 5μm using polymer beads. The liquid crystal (LC) was obtained from Merck (E-7 TN LC), and used as it was. In order to determine the direction of LC alignment, the dichroic dye (Disperse Blue 14) was used. It was dissolved 1% into E-7 LC. The dichroic dye shows strong absorption at 655nm. The absorption change as a function of polarization angle was measured UV-visible spectroscopy Photo-Alignment Using Polyimide Containing Methoxy
Cinnamate Derivatives
Su Young Kim1, Sung Eui Shin1 and Dong Myung Shin1
1Dept. of Chemical Engineering, Hongik University, Seoul 121-791, Korea
Tel.:82-2-320-1652, E-mail: [email protected]
P1-45 / S. Y. Kim
IMID 2009 DIGEST •
(HP model 8453) by rotating the polarizer.
Fig. 1. The polymerization of 3,5-diamino
benzyl alcohol and CBDA and surface reaction
of polyimide film and 4methoxy cinnamoyl
chloride
3. Results and discussion
Figure 2 shows 4-Methoxy cinnamic acid and 2-methoxy cinnamic acid have different isosbestic
points 268 and 250nm, respectively and 2,5-dimethoxy cinnamic acid has no isosbestic point.
4-methoxy cinnamic acid reacted most fast and
reached steady state at the lowest point. It
indicated that 4-methoxy cinnamic acid is
photo-sensitive and provide good alignment properties.
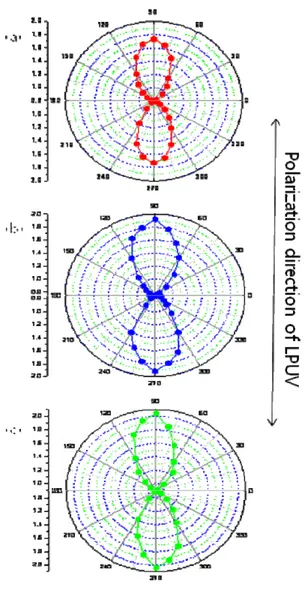
Polar diagrams (fig. 2) confirm direction of alignment. Polyimide films exposed to linearly polarized UV (LPUV) irradiation give homogenous LC alignment perpendicular to the polarization direction of the light. Table 1 indicate that LC cell with polyimide containion 4-methoxy cinnamate have highest pretilt angle. 4-Methoxy cinnamate effets on LC alignment efficiently.
Fig. 2. UV-visible absorption spectra of (a) 4-methoxy cinnamic acid, (b) 2-4-methoxy cinnamic acid and (c) 2,5- dimethoxy cinnamic acid in chloro-form irradiated with UV light with irradiation time
NH2 H2N OH O O O O O O + COOH HOOC O O HN OH HN * * n 3,5-diaminobenzyl alcohol CBDA
0~10°C NMP 210°C polyamic acid Cl O N N O O O O * * OH N N O O O O * * O O + n-Hexane Cat.:triethylamine O O surface reaction
polyimide 4-methoxy cinnamoyl chloride
2 4 0 2 6 0 2 8 0 3 0 0 3 2 0 3 4 0 3 6 0 3 8 0 4 0 0 0 .0 0 .2 0 .4 0 .6 0 .8 1 .0 A bs or ba nc e W a v e le n g th non-irrad iation 30s 1m in 2m in 3m in 5m in 7m in 10m in 15m in 20m in 25m in 30m in 35m in 40m in 45m in 2 5 0 3 0 0 3 5 0 4 0 0 0 .0 0 .2 0 .4 0 .6 0 .8 1 .0 A bs or ba nc e W avelength non 30s 1m in 2m in 3m in 5m in 7m in 10m in 15m in 20m in 25m in 30m in 4 0 0 0 .0 0 .1 0 .2 0 .3 0 .4 0 .5 0 .6 0 .7 0 .8 0 .9 1 .0 1 .1 A bs or ba nc e W avelength non 30s 1m in 2m in 3m in 5m in 7m in 10m in 15m in 20m in 25m in 30m in 35m in 40m in 45m in
P1-45 / S. Y. Kim
• IMID 2009 DIGEST
Fig. 3. Polar diagrams fo absorptions of a linearly polarized visible light (655nm wavelength) measured from LC cells fabricated with polyimide containing (a) 4-methoxy cinnamate, (b) 2-methoxy cinnamate, and (c)2,5- dimethoxy cinnamate film irradiated with LPUV as a function of the angle of rotation of the LC cell.
TABLE 1. Pretilt angle of LC cell with polyimide containing 4-methoxy cinnamate, 2-methoxy cinnamate and 2,5-dimethoxy cinnamate.
4-methoxy
cinnmate 2-methoxy cinnamte 2,5-dimethoxy cinnamate Pretilt
angle 2.78 1.183 0.356
4. Summary
The polyimide containing photo-reactive
cinnamate moiety as a side chain was
synthesized. Photo-alignment layer was
induced by irradiation with LPUV light. The
photo-sensitive for photo-alignment of
various photo-reactive molecules have been
studied by using UV-visible spectroscopy.
4-methoxy cinnamic acid reacted most faster
than others. It indicated that 4-methoxy
cinnamic acid is most photo-sensitive and
provide good alignment properties.
Homogenous LC alignment perpendicular to the polarization direction of the lightwas observed by
polar diagram and 4-methoxy has highest
pretilt angle. We considered 4-methoxy has
possibility for a good LC alignment
Acknowledgement
This work was supported by Ministry of Education, Science and Technology (2009-0071137)
5. References
1. K. Ichimura, V.P. Shibaev, Springer. 138. (1996) 2. P.J. Schannon, W.M. Gibbons, S.T. Sun, Nature 368
(1994) 532.
3. W.M. Gibbons, P.J. Schannon, S.T. Sun, B.J. Swetlin, Nature 351 (1991) 49.
4. M. Schadt, K. Schmitt, V. Kozinkov, V. Chigrinov, Jpn. J. Appl. Phys. 31 (1992)
5. M. Schadt, H. Seiberle, A. Schuster, Nature 381 (1996) 212.
6. Kawanishi, Y., Tamaki, T., Seki, T., Sakuragi, M., & Ichimura, K. Mol. Cryst. Liq. Cryst. Sci. Technol. Sect. A, 218, 153. (1992).
7. Hasegawa, M. & Taira, Y. J. Photopolym. Sci. Technol., 8, 241. (1995).
8. Sakamoto, K., Usami, K., Watanabe, W., Arafune, R., & Ushioda, S. Appl. Phys. Lett., 72, 1832.
(1998).
9. Newsome, C. J. & O’Neill, M. J. Appl. Phys., 92, 1752. (2002).
10. Matsuie, N., Ouchi, Y., Oji, H., Ito, E., Ishii, H., Seki, K., Hasegawa, M., & Zharnikov, M. Jpn. J. Appl. Phys., Part 2, 42, L67. (2003).
P1-45 / S. Y. Kim
IMID 2009 DIGEST •
11. Moon Hyung Cho, Kil Yeong Choi, Mi Hie Yi, Dong-Myung Shin. Colloids and Surfaces, 284, 267-269 (2006)
12. A. Dyadyusha, V. Kozinkov, T. Marusii, Y. Rcznikov, V. Rcshctnyak, A. Khizhnyak, Ukr. Fiz. Zh. 1059. 36. (1991).