Introduction
Introduction
Parkinson’s disease is a common neurodegenerative disease that occurs in about 1 in 100 elderly people 65 years of age or older [1]. It is characterized by the selective loss of substantia nigra dopaminergic neurons [1]. The most widely accepted mechanism of dopaminergic cell death in Parkinson’s disease
is the vicious cycle of oxidative stress [1,2]. Hydrogen peroxide (H2O2) is a key reactive oxygen species (ROS) and can induce
cell injury in a variety of cell types [3,4]. Recently, naturally oc-curring antioxidants are receiving great attention as they are recognized as safe and functional compounds for treating neu-rodegenerative diseases including Parkinson’s disease [1].
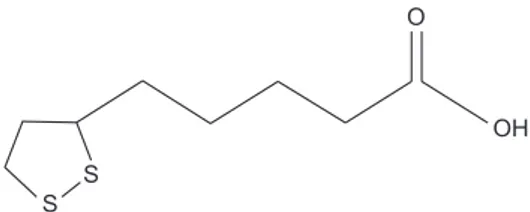
Alpha-lipoic acid (ALA, Fig. 1), a naturally occurring antioxi-Int J Oral Biol 46:15-22, 2021
pISSN: 1226-7155 • eISSN: 2287-6618 https://doi.org/10.11620/IJOB.2021.46.1.15
Alpha-lipoic acid protects human dopaminergic
neuronal cells against hydrogen peroxide-induced cell
injury by inhibiting autophagy and apoptosis
Kyeong-Rok Kang
1, Jae-Sung Kim
1, Tae-Hyeon Kim
1, Jeong-Yeon Seo
2, HyangI Lim
1, Jong-Hyun Park
1,
Kwang Yeol Yang
1, Sun-Kyoung Yu
1, Heung-Joong Kim
1, Chun Sung Kim
1, Hong Sung Chun
2, Dong-Seol Lee
3,
Joo-Cheol Park
3, and Do Kyung Kim
1*
1The Institute of Dental Science, Chosun University, Gwangju 61452, Republic of Korea
2Department of Integrative Biological Sciences & BK21 FOUR Educational Research Group for Age-associated Disorder Control
Technology, Chosun University, Gwangju 61452, Republic of Korea
3Laboratory for the Study of Regenerative Dental Medicine, Department of Oral Histology-Developmental Biology, School of Dentistry
and Dental Research Institute, Seoul National University, Seoul 08826, Republic of Korea
Alpha-lipoic acid (ALA) is a naturally occurring antioxidant and has been previously used to treat diabetes and cardiovascular disease. However, the autophagy effects of ALA against oxidative stress-induced dopaminergic neuronal cell injury remain unclear. The aim of this study was to investigate the role of ALA in autophagy and apoptosis against oxidative stress in the SH-SY5Y human dopaminergic neuronal cell line. We examined SH-SY5Y phenotypes using the 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide assay (cell viability/proliferation), 4′,6-diamidino-2-phenylindole dihydrochloride nuclear staining, Live/Dead cell assay, cellular reactive oxygen species (ROS) assay, immunoblotting, and immunocytochemistry. Our data showed ALA attenuated hydrogen peroxide (H2O2)-induced ROS generation and cell death. ALA effectively suppressed Bax up-regulation and 2 and
Bcl-xL down-regulation. Furthermore, ALA increased the expression of the antioxidant enzyme, heme oxygenase-1. Moreover, the expression of Beclin-1 and LC-3 autophagy biomarkers was decreased by ALA in our cell model. Combined, these data suggest ALA protects human dopaminergic neuronal cells against H2O2-induced cell injury by
inhibiting autophagy and apoptosis.
Keywords: Alpha-lipoic acid, Autophagy, Hydrogen peroxide, Neuronal cell
Received February 24, 2021; Revised March 10, 2021; Accepted March 10, 2021
*Correspondence to: Do Kyung Kim, E-mail: [email protected] https://orcid.org/0000-0001-6254-946X
Copyright © The Korean Academy of Oral Biology
CC This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License
(http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
dant, has been used to treat diabetes and cardiovascular dis-ease [5-7]. In particular, ALA is easily absorbed into the blood-stream and can easily cross through the blood-brain barrier, so it has been suggested that it can protect the central nervous system [8,9].
Autophagy, a different type of self-destructive process from apoptosis, is a major cellular pathway essential for cell sur-vival [10,11]. This process removes cellular waste products, degenerative proteins and degraded cell organelles [12,13]. The proteins and cell organelles to be removed are seques-tered into a vesicle called an autophagosome made of a double membrane, and this vesicle is bound to the lysosome to form an autophagolysosome [12,13]. These are broken down by ly-sosomal enzymes, and the broken down substances are used for the energy needed to survive the cell or for the creation of new organelles [12,13]. In other words, autophagy is one of the recycling systems in the cell [12,13]. Although the cyto-protective role of autophagy has been demonstrated in various experimental models, autophagy has also been reported to induce or participate in cell injury in certain circumstances [12-15].
Therefore, the purpose of this study is to investigate the role of ALA on autophagy and apoptosis against oxidative stress in the SH-SY5Y human dopaminergic neuronal cells, and to reveal the protective effect of ALA on dopamine neurons. Ultimately, we would like to present the efficacy of ALA as a treatment for degenerative brain diseases including Parkin-son’s disease. The SH-SY5Y cells are widely used to study the dopaminergic etiology due to represent a typical dopami-nergic markers, such as tyrosine hydroxylase and dopamine transporters [1,4,16]. So that, the SH-SY5Y cells used in this study could be suitable model cells to study the role of ALA on H2O2-mediated dopaminergic cell injury. Our results showed
that ALA protects human dopaminergic neuronal cells against H2O2-induced cell injury by inhibiting autophagy and apoptosis.
Materials and Methods
Materials and Methods
1. Materials
ALA (Fig. 1), 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltet-razolium bromide (MTT), H2O2, 4′,6-diamidino-2-phenylindole
dihydrochloride (DAPI), 2′,7′-Dichlorodihydrofluorescein di-acetate (H2DCFDA) were purchased from Sigma (St. Louis,
MO, USA). The Live/Dead cell viability assay kit was purchased from Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Anti-bodies against Bcl-2, Bcl-xL, Bax, heme oxygenase-1 (HO-1), Actin, and Beclin-1 were obtained from Santa Cruz Biotech-nology Inc. (Santa Cruz, CA, USA). The antibody against LC-3 was obtained from MBL Inc. (Woburn, MA, USA).
2. Cell culture and cell treatments
The human dopaminergic nuronal cell line, SH-SY5Y, was cultured in Dulbecco’s Modifed Eagls’s Medium/Nutrient Mix-ture F-12 (DMEM/F12, Thermo Scientific, Rockfrod, IL, USA) supplemented with 10% fetal bovine serum (Invitrogen, Carls-bad, CA, USA) and penicillin (100 U/mL)-streptomycin (100 µg/mL) at 37℃ in 5% CO2. When indicated, ALA was added
30 minutes prior to the treatment of H2O2. To prevent the
di-rect interaction between ALA and H2O2 in the culture medium,
at the end of the ALA pretreatment, the medium was changed to fresh low-serum DMEM/F12 medium.
3. Cytotoxicity assay
SH-SY5Y cells were seeded at a concentration of 1 × 104
cells per well in 48-well plates. After 24 hours growth, the cells were treated with ALA or H2O2 at various concentrations
for 24 hours. The cell viability test was evaluated using the MTT assay. At least 3 separate experiments were performed on each concentration combination. The supernatant was sub-sequently removed, and MTT crystals were dissolved in 200 µL/well dimethyl sulfoxide. Thereafter, optical density was measured at 570 nm using a spectrometer.
4. Cell Live/Dead assay
SH-SY5Y cell survival was measured using green calcein AM and ethidium homodimer-1, which stain live and dead cells, respectively. To evaluate cell survival, SH-SY5Y cells were plated on chamber slides, stimulated with H2O2 for 24 hours
OH O
S S
with or without pretreatment with ALA for 30 minutes, and then stained with green calcein AM and ethidium homodi-mer-1 according to the manufacturer’s protocol. Cells were then examined and imaged using fluorescence microscopy (Eclipse TE200; Nikon Instruments, Melville, NY, USA).
5. DAPI staining
SH-SY5Y cells were cultured in chamber slides at a seeding density of 1 × 104 cells per well for 24 hours, and then treated
with H2O2 for 24 hours with or without pretreatment with ALA
for 30 minutes. The treated SH-SY5Y cells were fixed with 4% paraformaldehyde (in phosphate buffered saline, PBS) for 15 minutes at room temperature and washed with PBS. The fixed cells were stained with DAPI (300 nM) for 15 minutes at room temperature in dark, washed with PBS and examined under fluorescent microscopy (Eclipse TE2000).
6. Measurement of intracellular ROS
SH-SY5Y cells were cultured in chamber slides at a seeding density of 1 × 104 cells per well for 24 hours, and then treated
with H2O2 for 24 hours with or without pretreatment with ALA
for 30 minutes. And then loaded with 20 µM H2DCFDA for 30
minutes at 37℃. Then cells were washed twice with phenol red-free DMEM/F12 and were incubated with H2O2 for 30
minutes. Cells were then examined and imaged using fluores-cence microscopy (Eclipse TE200).
7. Immunoblotting
The SH-SY5Y cells were treated with H2O2 for 24 hours with
or without pretreatment with ALA for 30 minutes. Immunob-lotting was done according to the previously described method with minor modifications [17]. The anti-Bcl-2, anti-Bcl-xL, anti-Bax, anti-HO-1, anti-Beclin-1, anti-LC-3, or anti-β-actin antibody was used as the primary antibody.
8. Haematoxylin and eosin staining
SH-SY5Y cells were cultured in chamber slides at a seeding density of 1 × 104 cells per well for 24 hours, and then treated
with H2O2 for 24 hours with or without pretreatment with ALA
for 30 minutes. Subsequently, the cells were fixed with 4% paraformaldehyde for 15 minutes at room temperature and washed with PBS. Haematoxylin and eosin staining were
per-formed to evaluate the morphological alterations in SH-SY5Y cells. Cells were observed and imaged using Leica DM 750 microscope (Leica Microsystems, Heerbrugg, Switzerland).
9. Immunocytochemistry
SH-SY5Y cells were cultured in chamber slides at a seeding density of 1 × 104 cells per well for 24 hours, and then treated
with H2O2 for 24 hours with or without pretreatment with ALA
for 30 minutes. Subsequently, the cells were fixed with 4% paraformaldehyde for 15 minutes at room temperature and washed with PBS. Immunocytochemistry was performed us-ing Vetastain ABC Kit (Vector Laboratories, Burlus-ingame, CA, USA). Fixed cell were incubated with Beclin-1 antibody for 1 hour and incubated with peroxidase-conjugated goat anti-mouse antibody for 1 hour. Cells were observed and imaged using Leica DM 750 microscope (Leica Microsystems).
10. Statistical analysis
All experiments were performed at least 3 times. The results were presented as mean ± standard deviation. The statistical significance was analyzed by using Student’s t-test for two groups and one way analysis of variance for multi-group com-parisons. All statistical analyses were performed using SPSS version 12.0 (SPSS Inc., Chicago, IL, USA). p-values < 0.05 were considered statistically significant.
Results
Results
1. ALA protects SH-SY5Y cells against H
2O
2-induced
cytotoxicity
In this study, the effect of ALA on H2O2-induced SH-SY5Y
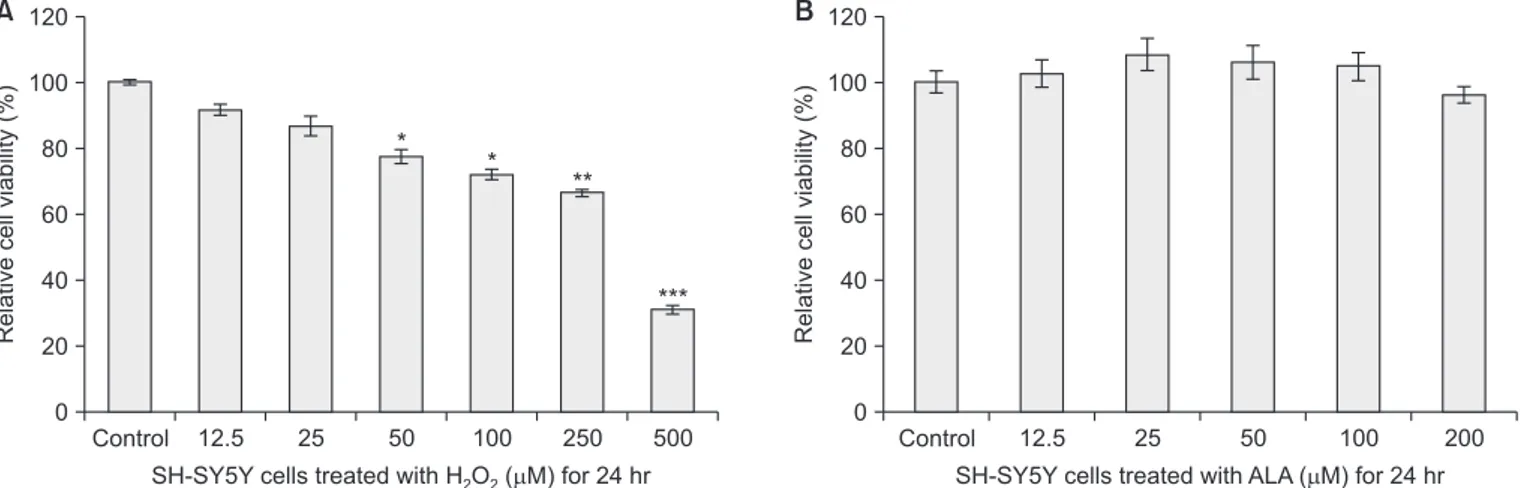
cell viability loss was assessed by MTT assay and Live/Dead assay. Treatment with 12.5–500 µM H2O2 decreased the
vi-ability of SH-SY5Y cells compared with the control in a dose-dependent manner (Fig. 2A). H2O2 (250 µM) induced
approxi-mately 35% cell loss after 24 hours treatment (Fig. 2A). SH-SY5Y cells were treated with 12.5–200 µM ALA for 24 hours, and an MTT assay was performed to verify if cell viability was altered. As shown in Fig. 2B, the viability of SH-SY5Y cells treated with 12.5, 25, 50, 100, and 200 µM ALA were 102 ± 4%, 108 ± 5%, 105 ± 3%, 104 ± 5%, and 96 ± 2%, respec-tively. These data indicate that ALA did not affect the cell vi-ability.
To investigate whether ALA could protect against H2O2
-in-duced dopaminergic cell death, SH-SY5Y cells were pretreated with 25 and 50 µM ALA for 30 minutes, followed by 250 µM H2O2 treatment for 24 hours. As shown in Fig. 3A, H2O2
-induced cell loss was attenuated by ALA treatment. To verify the survival of SH-SY5Y cells treated with ALA, cell Live/Dead assay using green calcein AM and ethidium homodimer-1 was performed. Cell Live/Dead assay showed an increase in the living population of SH-SY5Y cells incubated with ALA as compared with H2O2 treated cells (Fig. 3B upper panel). These
results indicated that ALA protects human dopaminergic
neu-2. ALA inhibited H
2O
2-induced apoptosis and ROS
production in SH-SY5Y cells
The nuclear condensation changes were assessed using DAPI staining. A significant proportion of SH-SY5Y cells with condensed nuclei increased upon exposure to H2O2, which
are the characteristics of apoptosis (Fig. 3B lower panel). The number of SH-SY5Y cells with condensed nuclei decreased by pretreatment with ALA in a dose-dependent manner.
To identify the changes of intracellular ROS in SH-SY5Y cells during the H2O2-induced cell death and ALA mediated
protec-A B Control 120 100 80 60 40 20 Relative cell viability (%)
SH-SY5Y cells treated with H O ( M) for 24 hr2 2
0 * ** *** 12.5 25 50 100 250 500 * Control 120 100 80 60 40 20 Relative cell viability (%)
SH-SY5Y cells treated with ALA ( M) for 24 hr 0
12.5 25 50 100 200
Fig. 2. Effects of hydrogen peroxide (H2O2) and alpha-lipoic acid (ALA) on cell cytotoxicity in SH-SY5Y human dopaminergic neuronal cells. The SH-SY5Y cells were treated with various concentrations of (A) H2O2 and (B) ALA for 24 hours. The cell viabilities were measured using an 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide assays. The percentage of cell viability was calculated as a ratio of A570nms of material treated cells and untreated con-trol cells. Each data point represents the mean ± standard deviation of three experiments.
*p < 0.05 vs. control, **p < 0.01 vs. control, and ***p < 0.001 vs. control (the control cells measured in the absence of H2O2 and ALA).
Control 120 100 80 60 40 20 Relative cell viability (%) H O (250 M)2 2 0 ** A B ALA 25 M ALA 50 M Live/Dead DAPI stain 0 25 50 H O (250 M) + ALA ( M) for 24 hr2 2 ** 100 M 100 M 100 M 100 M 100 M 100 M 100 M 100 M
Fig. 3. Cell death induced by hydrogen peroxide (H2O2) and protective effect of alpha-lipoic acid (ALA) in SH-SY5Y cells. (A) Indicated concentrations of ALA were added to the SH-SY5Y cells for 30 minutes before the addition of H2O2 (250 µM). Cell viability was assessed by 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide assay. Each data point represents the mean ± standard deviation of three experiments. (B) Cell Live/Dead assay (upper panel) and 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI) staining (lower panel) were performed to investigate the cell survival and chromatin condensation, respectively. The scale bar presented in each image is 100 µm.
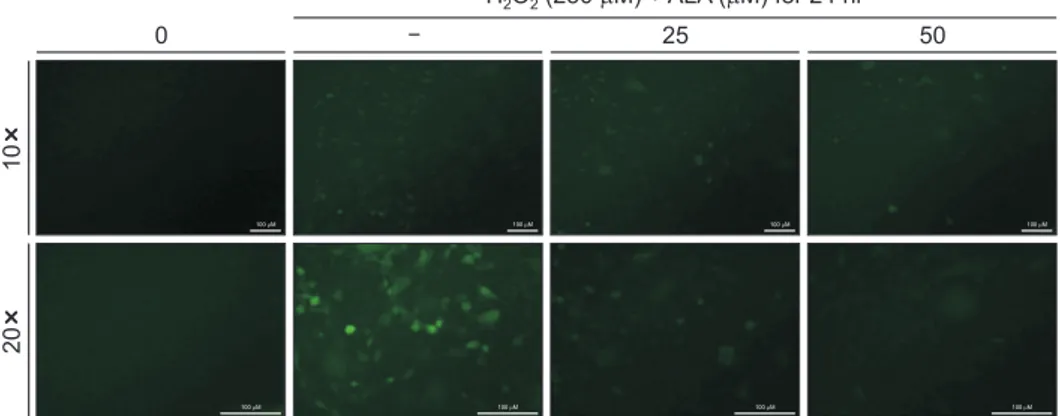
fluorescent dye H2DCFDA. As shown in Fig. 4, the levels of
fluorescence H2DCFDA increased after 30 minutes treatment
with H2O2. However, ALA decreased H2O2-induced ROS
pro-duction in a ALA dose dependent-manner (Fig. 4).
3. ALA protects H
2O
2-induced cell death through
down-regulating oxidative stress
Immunoblotting was performed to verify if the expres-sions of anti-apoptotic Bcl-2, Bcl-xL and pro-apoptotic Bax are modulated by ALA. After treatment with 250 µM H2O2 for
24 hours, the expression of Bcl-2 and Bcl-xL was markedly decreased but the expression of Bax was increased (Fig. 5A). However, pretreatment with ALA decreased the expression of Bax and increased the expression level of Bcl-2 and Bcl-xL in SY5Y cells (Fig. 5A). To confirm the oxidative stress in SH-SY5Y cells during the H2O2-induced cell death and ALA
medi-ated protection, the alteration of biomarker associmedi-ated with inducible enzyme with antioxidant was investigated. As shown
in Fig. 5B, ALA significantly increased the expression of HO-1, biomarker associated with antioxidant. These data indicate that ALA protects H2O2-induced dopaminergic cell death through
down-regulating apoptosis and oxidative stress.
4. ALA inhibits autophagy in H
2O
2-induced SH-SY5Y
cell injury
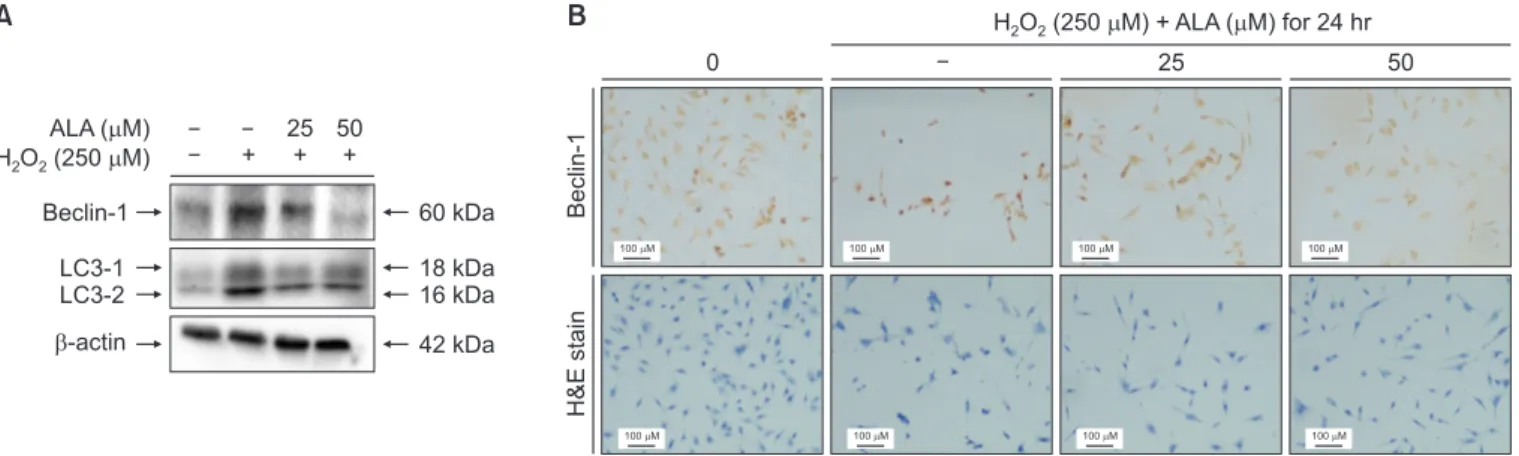
To determine whether the autophagic processes is involved in H2O2-induced cell death, immunoblotting and
immuno-cytochemistry were performed. As shown in Fig. 6A, after treatment with 250 µM H2O2 for 24 hours, the expressions of
Beclin-1 and LC-3, the specific biomarkers associated with autophagy, were markedly increased. However, pretreatment with ALA decreased the expression levels of Beclin-1 and LC-3 in SH-SY5Y cells (Fig. 6A). Futhermore, the immunore-activity of Beclin-1 decreased by 25 and 50 µM pretreatment with ALA in SH-SY5Y cells, as indicated by the results of im-munocytochemistry (Fig. 6B). These results indicate that ALA
ALA ( M) H O (2502 2 M) Bcl-2 Bcl-xL Bax -actin + 25 + 50 + 26 kDa 30 kDa 20 kDa 42 kDa ALA ( M) H O (2502 2 M) HO-1 -actin + 25 + 50 + 32 kDa 42 kDa A B
Fig. 5. Effects of alpha-lipoic acid (ALA) on hydrogen peroxide (H2O2)-induced oxidative stress in SH-SY5Y cells. (A) Expressions of anti-apoptotic factors Bcl-2, Bcl-xL, and pro-apoptotic factor Bax in SH-SY5Y cells. (B) Expression of heme oxygenase-1 (HO-1) in SH-SY5Y cells. SH-SY5Y cells were treated with H2O2 (250 µM) for 24 hours. ALA was added to the media for 30 minutes before the addition of H2O2. The cell lysate was prepared and analyzed by im-munoblotting as described in “Materials and Methods”.
Fig. 4. Effects of alpha-lipoic acid (ALA) on hydrogen peroxide (H2O2)-induced reactive oxygen species production in SH-SY5Y cells. Cells were cultured in chamber slides at a seeding density of 1 × 104 cells per well for 24 hours, and then treated with H
2O2 for 24 hours with or without pretreatment with ALA for 30 minutes. And then loaded with 20 µM 2′,7′-Dichlorodihydrofluorescein diacetate for 30 minutes at 37℃. Images were observed by fluorescence micros-copy (Eclipse TE2000; Nikon Instruments, Melville, NY, USA). The scale bar presented in each image is 100 µm.
10 20 0 25 50 H O (250 M) + ALA ( M) for 24 hr2 2 100 M 100 M 100 M 100 M 100 M 100 M 100 M 100 M
inhibits autophagy in H2O2-induced SH-SY5Y cells.
Discussion
Discussion
ALA, which is a potent antioxidant is easily absorbed into the bloodstream and can without difficulty cross through the blood-brain barrier, so it has been advised that it can be protect the central nervous system [8,9]. SH-SY5Y cells are widely used to in the study dopaminergic pathogenesis [1,4,16]. This is because these cell line represent several typical dopaminergic pathological markers, such as tyrosine hydroxylase and dopa-mine transporter [1,4,16]. In this study, the protective effect of ALA in H2O2-induced cell death was examined.
MTT assay and cell Live/Dead assay showed that ALA did not affect the cell viability and survival of SH-SY5Y cells. These data suggested that ALA has been nontoxicity to SH-SY5Y cells. In addition, we found that ALA could regulate H2O2
-induced cell death in SH-SY5Y human dopaminergic neuronal cells (Figs. 2 and 3).
It is well accepted that ROS may attack intracellular biological macromolecules, including nuclear acids, proteins and lipids, dysfunction of mitochondria, and activate signaling pathways leading to apoptosis [18,19]. In this study, our results showed the apoptotic activity, pro-apoptotic Bax up-regulation and an-ti-apoptotic Bcl-2 and Bcl-xL down-regulations when treated with H2O2 in SH-SY5Y cells (Figs. 3 and 5). Interestingly, ALA
pretreatment reduced the formation of nuclear condensa-tion and decreased the levels of Bax protein expression, but increase the levels of Bcl-2 and Bcl-xL protein expressions in
that ALA exerted its protective effects through ROS reduction (Fig. 4). These data suggest that ALA protects H2O2-induced
cell injury through down-regulating apoptosis and oxidative stress.
Several studies reported that HO-1 has neuroprotective ef-fects against oxidative stress-induced neuronal damage [20-22]. Although HO-1 protein levels are normally low in neurons, HO-1 can be highly up-regulated in cerebral ischemia [23] and during the formation of neurofibrillary tangles in Alzheimer’ s disease [24] and markedly accumulated in neuronal Lewy bodies of Parkinson’s patients [25]. Heme and other oxidative stress stimuli enhance the HO-1 expression in practically all tissues and cells including neurons [25]. Similar to other previ-ous studies [20-25], in this study, the pretreatment with ALA increased HO-1 expression in SH-SY5Y cells (Fig. 5). These data indicate that ALA may increase cell resistance to oxidative stress and delay the dopaminergic cell injury.
Autophagy is an important physiological process that de-grades intracellular components, such as cellular waste prod-ucts, degenerative proteins and damaged cellular organelles [12,13]. Beclin-1 and LC-3 are common biomarkers of au-tophagy [14]. Auau-tophagy may be a mechanism for cell survival but may also induce cell death [12]. At the early stage of cell stress, autophagy is induced and then cytoprotective effect [12]. When cell stress is too severe at late stage, exces-sive autophagy may trigger cell injury and death [12]. Hence, to verify whether autophagic processes is involved in H2O2
-induced cell injury, the alteration of biomarkers associated with autophagy was investigated. In our study, the expressions
B Beclin-1 H&E stain 0 25 50 H O (250 M) + ALA ( M) for 24 hr2 2 A ALA ( M) H O (2502 2 M) Beclin-1 LC3-1 LC3-2 -actin + 25 + 50 + 60 kDa 18 kDa 16 kDa 42 kDa 100 M 100 M 100 M 100 M 100 M 100 M 100 M 100 M
Fig. 6. Effect of alpha-lipoic acid (ALA) on autophagic processes in hydrogen peroxide (H2O2)-induced cell injury. (A) Immunoblotting was performed to veri-fy the expression of Beclin-1 and LC-3, the specific biomarkers associated with autophagy. (B) Immunocytochemistry was performed to veriveri-fy the expression of Beclin-1. SH-SY5Y cells were treated with H2O2 (250 µM) for 24 hours. ALA was added to the media for 30 minutes before the addition of H2O2. Cells were observed and imaged using Leica DM 750 microscope (Leica Microsystems, Heerbrugg, Switzerland).
in SH-SY5Y cells. These findings indicate that ALA attenu-ated the autophagic processes in H2O2-induced dopaminergic
cell injury (Fig. 6). On the other hand, the mechanisms of cell protective effects induced by ALA in dopaminergic neuronal cells were not fully understood [26]. Therefore, further studies are needed to investigate the precise cellular and molecular mechanisms of cell apoptosis induced by ALA.
In conclusion, these results suggest that ALA protects SH-SY5Y human dopaminergic neuronal cells against H2O2
-induced cell injury by inhibiting autophagy and apoptosis. Sub-sequent studies will allow us to elucidate that ALA may be a promising candidate for the treatment and prevention of
treat-ing neurodegenerative diseases includtreat-ing Parkinson’s disease.
Acknowledgements
Acknowledgements
This research was supported by HysensBio Co., Ltd funded (2020).
Conflicts of Interest
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
References
References
1. Lee HJ, Cho HS, Park E, Kim S, Lee SY, Kim CS, Kim DK, Kim SJ, Chun HS. Rosmarinic acid protects human dopaminergic neuronal cells against hydrogen peroxide-induced apoptosis. Toxicology 2008;250:109-15. doi: 10.1016/j.tox.2008.06.010. 2. Zhang Y, Dawson VL, Dawson TM. Oxidative stress and ge-netics in the pathogenesis of Parkinson’s disease. Neurobiol Dis 2000;7:240-50. doi: 10.1006/nbdi.2000.0319.
3. Jenner P. Oxidative stress in Parkinson’s disease. Ann Neurol 2003;53 Suppl 3:S26-36; discussion S36-8. doi: 10.1002/ ana.10483.
4. Wang W, Huang W, Li L, Ai H, Sun F, Liu C, An Y. Morroni-side prevents peroxide-induced apoptosis by induction of endogenous glutathione in human neuroblastoma cells. Cell Mol Neurobiol 2008;28:293-305. doi: 10.1007/s10571-007-9168-7.
5. Ziegler D, Reljanovic M, Mehnert H, Gries FA. Alpha-lipoic acid in the treatment of diabetic polyneuropathy in Germany: current evidence from clinical trials. Exp Clin Endocrinol Dia-betes 1999;107:421-30. doi: 10.1055/s-0029-1212132. 6. Wollin SD, Jones PJ. Alpha-lipoic acid and cardiovascular
disease. J Nutr 2003;133:3327-30. doi: 10.1093/jn/133.11. 3327.
7. Park E, Gim J, Kim DK, Kim CS, Chun HS. Protective effects of alpha-lipoic acid on glutamate-induced cytotoxicity in C6 glioma cells. Biol Pharm Bull 2019;42:94-102. doi: 10.1248/ bpb. b18-00603.
8. Astiz M, de Alaniz MJ, Marra CA. The oxidative damage and inflammation caused by pesticides are reverted by lipoic acid in rat brain. Neurochem Int 2012;61:1231-41. doi: 10.1016/j. neuint.2012.09.003.
9. Yang TY, Xu ZF, Liu W, Xu B, Deng Y, Li YH, Feng S.
Alpha-lipoic acid protects against methylmercury-induced neurotoxic effects via inhibition of oxidative stress in rat cerebral cortex. Environ Toxicol Pharmacol 2015;39:157-66. doi: 10.1016/j. etap.2014.11.020.
10. Onodera J, Ohsumi Y. Autophagy is required for maintenance of amino acid levels and protein synthesis under nitrogen starvation. J Biol Chem 2005;280:31582-6. doi: 10.1074/jbc. M506736200.
11. Hu J, Cui W, Ding W, Gu Y, Wang Z, Fan W. Globular adipo-nectin attenuated H2O2-induced apoptosis in rat chondro-cytes by inducing autophagy through the AMPK/ mTOR path-way. Cell Physiol Biochem 2017;43:367-82. doi: 10.1159/ 000480416.
12. Periyasamy-Thandavan S, Jiang M, Schoenlein P, Dong Z. Autophagy: molecular machinery, regulation, and implications for renal pathophysiology. Am J Physiol Renal Physiol 2009; 297:F244-56. doi: 10.1152/ajprenal.00033.2009.
13. Mariño G, Niso-Santano M, Baehrecke EH, Kroemer G. Self-consumption: the interplay of autophagy and apoptosis. Nat Rev Mol Cell Biol 2014;15:81-94. doi: 10.1038/nrm3735. 14. Bursch W, Ellinger A, Gerner C, Fröhwein U,
Schulte-Her-mann R. Programmed cell death (PCD). Apoptosis, autophagic PCD, or others? Ann N Y Acad Sci 2000;926:1-12. doi: 10. 1111/j.1749-6632.2000.tb05594.x.
15. Galluzzi L, Morselli E, Vicencio JM, Kepp O, Joza N, Tajeddine N, Kroemer G. Life, death and burial: multifaceted impact of autophagy. Biochem Soc Trans 2008;36(Pt 5):786-90. doi: 10.1042/BST0360786.
16. Hasegawa T, Matsuzaki M, Takeda A, Kikuchi A, Furukawa K, Shibahara S, Itoyama Y. Increased dopamine and its metabo-lites in SH-SY5Y neuroblastoma cells that express tyrosinase.
J Neurochem 2003;87:470-5. doi: 10.1046/j.1471-4159.2003. 02008.x.
17. Cho JH, Kim SG, Park BS, Go DS, Park JC, Kim DK. MicroR-NA-27 promotes odontoblast differentiation via Wnt1 signal-ing. Int J Oral Biol 2015;40:197-204. doi: 10.11620/IJOB.2015. 40.4.197.
18. Chun HS, Lee H, Son JH. Manganese induces endoplasmic reticulum (ER) stress and activates multiple caspases in nigral dopaminergic neuronal cells, SN4741. Neurosci Lett 2001; 316:5-8. doi: 10.1016/s0304-3940(01)02341-2.
19. Barbouti A, Doulias PT, Nousis L, Tenopoulou M, Galaris D. DNA damage and apoptosis in hydrogen peroxide-exposed Jurkat cells: bolus addition versus continuous generation of H(2)O(2). Free Radic Biol Med 2002;33:691-702. doi: 10. 1016/s0891-5849(02)00967-x.
20. Ahmad AS, Zhuang H, Doré S. Heme oxygenase-1 protects brain from acute excitotoxicity. Neuroscience 2006;141:1703-8. doi: 10.1016/j.neuroscience.2006.05.035.
21. Barañano DE, Snyder SH. Neural roles for heme oxygenase: contrasts to nitric oxide synthase. Proc Natl Acad Sci U S A
2001;98:10996-1002. doi: 10.1073/pnas.191351298. 22. Ryter SW, Alam J, Choi AM. Heme oxygenase-1/carbon
monoxide: from basic science to therapeutic applications. Physiol Rev 2006;86:583-650. doi: 10.1152/physrev.00011. 2005.
23. Nimura T, Weinstein PR, Massa SM, Panter S, Sharp FR. Heme oxygenase-1 (HO-1) protein induction in rat brain fol-lowing focal ischemia. Brain Res Mol Brain Res 1996;37:201-8. doi: 10.1016/0169-328x(95)00315-j.
24. Takeda A, Itoyama Y, Kimpara T, Zhu X, Avila J, Dwyer BE, Perry G, Smith MA. Heme catabolism and heme oxygenase in neurodegenerative disease. Antioxid Redox Signal 2004;6: 888-94. doi: 10.1089/ars.2004.6.888.
25. Schipper HM. Heme oxygenase expression in human central nervous system disorders. Free Radic Biol Med 2004;37:1995-2011. doi: 10.1016/j.freeradbiomed.2004.09.015.
26. Jeong KI, Kim SG, Go DS, Kim DK. Inhibition of cell growth and induction of apoptosis by bilobalide in FaDu human pha-ryngeal squamous cell carcinoma. Int J Oral Biol 2020;45:8-14. doi: 10.11620/IJOB.2020.45.1.8.