Outcomes and recurrence pattern after non-anatomic liver resection for solitary hepatocellular carcinomas
Sung Hoon Cho, Jae Min Chun, Hyung Jun Kwon, Young Seok Han, Sang Geol Kim, and Yoon Jin Hwang
Department of Surgery, Kyungpook National University Hospital, Kyungpook National University School of Medicine, Daegu, Korea
Backgrounds/Aims: Anatomic resection (AR) is preferred for eradicating portal tributaries in patients with hepatocellular carcinoma (HCC). However, the extent of resection is influenced by underlying liver disease and tumor location. We compared the surgical outcomes and recurrence pattern between non-anatomic resection (NR) and AR. Methods: From March 2009 to February 2012, 184 patients underwent surgical resection for HCC. Among these, 79 patients who were primarily treated for a single tumor without rupture or macroscopic vascular invasion were enrolled. The patients were divided into 2 groups based on the extent of resection: AR (n=31) or NR (n=48). We compared the clinical charac- teristics, overall survival, disease-free survival, pattern of recurrence, and biochemical liver functions during the peri- operative period between the two groups. Results: The extent of resection had no significant effect on overall or dis- ease-free survival rates. The overall 1- and 3-year survival rates were 97% and 82% in the AR group, and 96% and 89% in the NR group, respectively (p=0.49). In addition, the respective 1- and 3-year disease-free survival rates for the AR and NR groups were 84% and 63%, and 85% and 65%, respectively (p=0.94). On the other hand, the presence of hepatic cirrhosis and a tumor size of >5 cm were significant risk factors for recurrence according to multivariate analysis (p<0.001 and p=0.003, respectively). The frequency of early recurrence, the first site of recurrence, and the pattern of intrahepatic recurrence were similar between the 2 groups (p=0.419, p=0.210, and p=0.734, respectively);
in addition, the frequency of marginal recurrence did not differ between the 2 groups (1 patient in the AR group and 2 in the NR group). The NR group showed better postoperative liver function than the AR group. Conclusions: Non-ana- tomic liver resection can be a safe and efficient treatment for patients with a solitary HCC without rupture or gross vascular invasion. (Korean J Hepatobiliary Pancreat Surg 2016;20:1-7)
Key Words: Hepatocellular carcinoma; Resection; Recurrence pattern; Disease-free survival
Received: May 28, 2015; Revised: September 26, 2015; Accepted: September 30, 2015 Corresponding author: Jae Min Chun
Department of Surgery, Kyungpook National University School of Medicine, 50 Samduk-dong 2 ga, Chung-gu, Daegu 41944, Korea Tel: +82-53-420-5605, Fax: +82-53-421-0510, E-mail: cjmdr@knu.ac.kr
Copyright Ⓒ 2016 by The Korean Association of Hepato-Biliary-Pancreatic Surgery
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/
licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Korean Journal of Hepato-Biliary-Pancreatic Surgery ∙ pISSN: 1738-6349ㆍeISSN: 2288-9213
INTRODUCTION
Hepatocellular carcinoma (HCC) is the fifth most com- mon malignancy worldwide, and South Korea has one of the highest prevalence rates for this disease.1 Surgical re- section is accepted as the most effective intervention to cure such patients. There are several reports on the risk factors for tumor recurrence after liver resection for HCC, mostly based on patient status, pathology results, and bio- logical factors, irrespective of the surgical precision or procedure adopted.2
When the extent and type of liver resection are consid- ered, anatomic resection (AR) is theoretically superior to non-anatomic resection (NR) for patients with HCC be-
cause it can eradicate intrahepatic metastases that might have spread via portal tributaries.3 However, the fear of further reducing the liver size causes surgeons to question the benefits of performing AR in a patient with cirrhosis.
Moreover, the superiority of this procedure for oncologic safety remains controversial.4,5 Therefore, we retro- spectively evaluated patient survival rates and recurrence patterns, and compared the post-hepatectomy liver func- tion with the extent of liver resection in such patients.
MATERIALS AND METHODS
From March 2009 to February 2012, 184 patients under- went curative surgical resection for HCC at Kyungpook
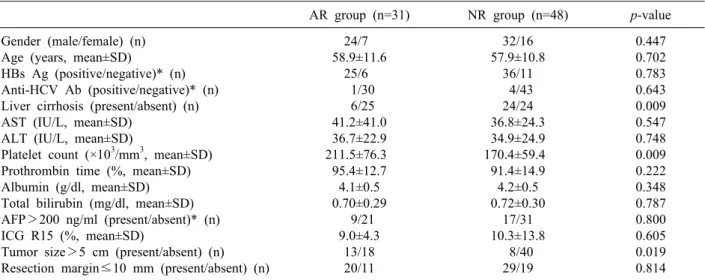
Table 1. Clinicopathological features of the anatomic and non-anatomic resection groups
AR group (n=31) NR group (n=48) p-value
Gender (male/female) (n) Age (years, mean±SD)
HBs Ag (positive/negative)* (n) Anti-HCV Ab (positive/negative)* (n) Liver cirrhosis (present/absent) (n) AST (IU/L, mean±SD)
ALT (IU/L, mean±SD)
Platelet count (×103/mm3, mean±SD) Prothrombin time (%, mean±SD) Albumin (g/dl, mean±SD) Total bilirubin (mg/dl, mean±SD) AFP>200 ng/ml (present/absent)* (n) ICG R15 (%, mean±SD)
Tumor size>5 cm (present/absent) (n) Resection margin≤10 mm (present/absent) (n)
24/7 58.9±11.6
25/6 1/30 6/25 41.2±41.0 36.7±22.9 211.5±76.3 95.4±12.7 4.1±0.5 0.70±0.29
9/21 9.0±4.3
13/18 20/11
32/16 57.9±10.8
36/11 4/43 24/24 36.8±24.3 34.9±24.9 170.4±59.4 91.4±14.9 4.2±0.5 0.72±0.30
17/31 10.3±13.8
8/40 29/19
0.447 0.702 0.783 0.643 0.009 0.547 0.748 0.009 0.222 0.348 0.787 0.800 0.605 0.019 0.814
*Data available for 79 patients. AR, anatomic resection; NR, non-anatomic resection; HBs Ag, hepatitis B surface antigen; an- ti-HCV Ab, anti-Hepatitis C virus antibody; AST, aspartate aminotransferase; ALT, alanine aminotransferase; AFP, -fetoprotein;
ICG R15, indocyanine green dye retention rate at 15 minutes National University Hospital. Among these, 79 who under- went surgical resection as a first-line therapy for a single tumor were enrolled in this study, excluding patients with macroscopic vascular invasion or a ruptured HCC. A cura- tive hepatic resection was defined as macroscopic removal of all tumorous tissue from the liver, and AR was defined as complete resection of the anatomical area that was identi- fied from preceding ischemia. There were 31 cases of AR, of which, 16 involved hemihepatectomy, 4 involved ex- tended hemihepatectomy, and 11 involved sectionectomy.
All the other procedures were classified as NR and per- formed in 48 patients. During the NR procedures, we fo- cused on creating a sufficient resection margin of 1 cm, regardless of the condition of the hepatic pedicle. Early recurrence was defined as a recurrence within 2 years after a curative resection, in accordance with previous studies.6,7 A serial follow-up of the patients was provided to monitor for any recurrence. Contrast-enhanced abdominal computed tomography (CT) scans were repeated every 3 months dur- ing the first 2 postoperative years and alternating abdominal ultrasound and abdominal CT scans were performed every 3 months thereafter. Serum -fetoprotein (AFP) levels were measured every 3 months. Recurrence patterns after liver resection were classified according to the location of the recurring tumor and the time to recurrence, and the pattern of intrahepatic recurrence was classified according to Poon et al.8 In addition, post-hepatectomy liver functions includ-
ing the circulating levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), total bilirubin, and prothrombin time were compared with the extent of liver resection during the perioperative period.
Student’s independent t tests were used to compare the differences between continuous variables in patients with and without AR. Categorical variables were subjected to chi-squared analysis if the sample size was adequate or Fisher’s exact test if the sample number was small. The cumulative overall and disease-free survival rates were calculated using the Kaplan-Meier method, and candidate variables with p-values <0.2 by univariate analysis were further analyzed using the Cox proportional-hazards model. The statistical analyses were performed using IBM SPSS statistics for Windows version 20.0 (IBM Corp., Armonk, NY, USA), and p<0.05 was considered statisti- cally significant. Results were presented as the hazard ra- tio (HR) and 95% confidence interval (CI).
RESULTS
Patient characteristics
The clinical features of the 79 patients were listed in Table 1. Comparing the AR group (n=31) versus the NR group (n=48), there were no significant differences in gen- der, age, the incidences of hepatitis B or C, AST or ALT levels, prothrombin time (PT), serum albumin, total bilir-
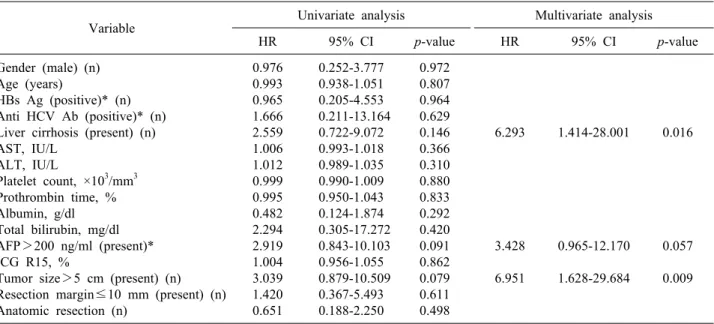
Table 2. Univariate and multivariate analysis of variables related to overall survival after hepatic resection for HCC
Variable Univariate analysis Multivariate analysis
HR 95% CI p-value HR 95% CI p-value
Gender (male) (n) Age (years)
HBs Ag (positive)* (n) Anti HCV Ab (positive)* (n) Liver cirrhosis (present) (n) AST, IU/L
ALT, IU/L
Platelet count, ×103/mm3 Prothrombin time, % Albumin, g/dl Total bilirubin, mg/dl AFP>200 ng/ml (present)*
ICG R15, %
Tumor size>5 cm (present) (n) Resection margin≤10 mm (present) (n) Anatomic resection (n)
0.976 0.993 0.965 1.666 2.559 1.006 1.012 0.999 0.995 0.482 2.294 2.919 1.004 3.039 1.420 0.651
0.252-3.777 0.938-1.051 0.205-4.553 0.211-13.164
0.722-9.072 0.993-1.018 0.989-1.035 0.990-1.009 0.950-1.043 0.124-1.874 0.305-17.272 0.843-10.103 0.956-1.055 0.879-10.509
0.367-5.493 0.188-2.250
0.972 0.807 0.964 0.629 0.146 0.366 0.310 0.880 0.833 0.292 0.420 0.091 0.862 0.079 0.611 0.498
6.293
3.428 6.951
1.414-28.001
0.965-12.170 1.628-29.684
0.016
0.057 0.009
*Data available for 79 patients. HR, Hazard ratio; CI, confidence interval; HBs Ag, hepatitis B surface antigen; anti-HCV Ab, anti-Hepatitis C virus antibody; AST, aspartate aminotransferase; ALT, alanine aminotransferase; AFP, -fetoprotein; ICG R15, indocyanine green dye retention rate at 15 minutes
Fig. 1. Cumulative survival curves of all 79 patients. The overall survival rates (A) and disease-free survival rates (B) were 97%, 86% at 1 year and 85%, 64% at 3 years, respectively.
ubin, AFP levels, indocyanine green dye retention rate at 15 min, or tumor-free resection margin. However, tumors were significantly larger in the AR group, as compared with the NR group (p=0.019); in addition, patients in the NR group had a significantly lower platelet count (p=0.009) and higher rates of hepatic cirrhosis (p=0.009), as compared with those in the AR group. There were no patients with Child-Pugh class B or C tumors in either group.
Recurrence and survival outcomes
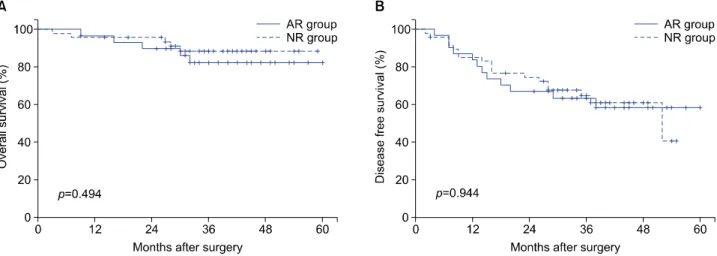
After a median follow-up of 37.0 months (range 3-60) post-resection, the 1- and 3-year overall and disease-free survival rates for all patients were 97% and 86%, and 85% and 64%, respectively (Fig. 1).
Thirty patients (38%) had recurrence and 10 patients died during the follow-up period. In the NR group, 4 pa- tients died of recurrent HCC, 1 died of hepatic failure, 14 patients remain alive with recurrent HCC, and 29 are alive and disease free. Among the patients in the AR group, 5 died of recurrent HCC, 7 remain alive with re-
Fig. 2. Survival curves showing the overall survival (A) and disease-free survival (B) after curative surgery for HCC in the anatomic resection (AR) group (solid line) and non-anatomic resection (NR) group (dotted line).
Table 3. Univariate and multivariate analysis of variables related to disease free-survival after hepatic resection for HCC
Variable Univariate analysis Multivariate analysis
HR 95% CI p-value HR 95% CI p-value
Gender (male) (n) Age (years)
HBs Ag (positive)* (n) Anti HCV Ab (positive)* (n) Liver cirrhosis (present) (n) AST, IU/L
ALT, IU/L
Platelet count, ×103/mm3 Prothrombin time, % Albumin, g/dl Total bilirubin, mg/dl
AFP>200 ng/ml (present)* (n) ICG R15, %
Tumor size>5 cm (present) (n) Resection margin≤10 mm (present) (n) Anatomic resection (n)
1.370 1.017 0.837 1.243 3.389 1.000 0.993 0.998 1.000 0.778 0.683 1.071 0.971 1.728 1.031 0.974
0.584-3.212 0.983-1.052 0.359-1.952 0.295-5.230 1.605-7.156 0.989-1.010 0.975-1.010 0.993-1.004 0.974-1.026 0.362-1.715 0.210-2.218 0.474-2.419 0.915-1.030 0.808-3.697 0.490-2.168 0.468-2.027
0.469 0.338 0.680 0.767 0.001 0.945 0.400 0.541 0.973 0.548 0.526 0.869 0.328 0.158 0.937 0.944
5.465
3.646
2.339-12.770
1.537-8.650
<0.001
0.003
*Data available for 79 patients. HR, Hazard ratio; CI, confidence interval; HBs Ag, hepatitis B surface antigen; anti-HCV Ab, anti-Hepatitis C virus antibody; AST, aspartate aminotransferase; ALT, alanine aminotransferase; AFP, -fetoprotein; ICG R15, indocyanine green dye retention rate at 15 minutes
current HCC, and 19 are alive and disease free.
Table 2 listed the prognostic factors associated with overall survival of these patients identified from univariate and multivariate analyses. In univariate analysis, the pres- ence of liver cirrhosis, an AFP level >200 ng/ml, and a tumor size of >5 cm showed p-values of <0.2 and were further included in multivariate Cox propor- tional-hazard regression analysis; the presence of liver cir- rhosis (HR 6.293; 95% CI 1.414-28.001; p=0.016) and tu- mor size >5 cm (HR 6.951; 95% CI 1.628-29.684;
p=0.009) were independent prognostic factors for overall
survival. On univariate analysis of the risk factors for tu- mor recurrence, the presence of liver cirrhosis and tumor size >5 cm showed p-values <0.2 and the presence of liver cirrhosis (HR 5.465; 95% CI 2.339-12.770; p<
0.001) and tumor size >5 cm (HR 3.646; 95% CI 1.537-8.650; p=0.003) were significant independent pre- dictors of tumor recurrence after liver resection (Table 3).
On the other hand, the extent of liver resection was not a significant risk factor for recurrence with the 1- and 3-year overall survival rates for the AR and NR groups of 97% and 82%, and 96% and 89%, respectively
Fig. 3. Biochemical liver function parameters of the anatomic resection (AR) group (solid line) and non-anatomic resection (NR) group during the perioperative period. Asterisks indicated p-value <0.05.
Table 4. Patterns of tumor recurrence following hepatic re- section for HCC
Extent of resection p-value AR group NR group Time until recurrence
≤2 years >2 years
First site of recurrence Intrahepatic
Extrahepatic
Pattern of intrahepatic recurrence Marginal
Adjacent section Distal section Multisectional
10 2
7 5 1 2 3 1
12 6
15 3 2 5 3 5
0.419
0.210
0.734
AR, anatomic resection; NR, non-anatomic resection (p=0.494), and the 1- and 3-year disease-free survival rates
of 84% and 63% in the AR group, and 85% and 65%
in the NR group (p=0.944; Fig. 2).
Patterns of tumor recurrence
Twenty-two patients (28%) had tumors that recurred within 2 years after liver resection (10 patients after AR and 12 after NR) and the frequency of early recurrences was similar between the 2 groups (p=0.419). Regarding the pattern of recurrence, in the NR group, 2 patients had a recurrence at the liver transection margin (1 of these showed a marginal recurrence at 36 months after liver re- section), 5 had a recurrence in an adjacent section, 3 in a distal section, 5 had a multi-sectional recurrence, and 3 developed extrahepatic recurrences. In the AR group, a marginal recurrence occurred in 1 patient. The other intra- hepatic recurrences consisted of 2 at an adjacent section, 3 at a distal section, and 1 in multiple sections. Five pa- tients had a recurrence at an extrahepatic site. There were
no significant differences between the 2 groups with re- spect to the first site of recurrence or the pattern of intra- hepatic recurrence (p=0.210 and p=0.734, respectively;
Table 4).
Perioperative liver function
Biochemical liver function parameters were similar be- tween the 2 groups during the preoperative period.
However, the serum bilirubin level and PT value of the AR group were worse than those of the NR group during the postoperative period (Fig. 3).
DISCUSSION
AR is theoretically ideal for a patient with HCC from functional and oncological aspects, because the related portal vein and corresponding hepatic territory are removed. This reduces the ischemic area, minimizes any bile leakage, and lessens the possibility of any local re- currence of the tumor. However, the removal of a larger liver volume in the AR vs. NR procedure raises the possi- bility that the remaining liver mass might not meet the patient’s metabolic requirements.9,10 Therefore, several studies have aimed to evaluate the effects of the extent of surgery for HCC.4,5,11-13 In 2014, 3 studies used propen- sity-matching score analysis with the aim of identifying any benefit in patient survival after AR. However, none of these found conclusive results despite efforts to mini- mize confounding factors; in fact, they reported three dif- ferent results. Okamura et al.14 showed that the overall and recurrence-free survival in patients with HCC infected with hepatitis C virus were not superior in AR than NR procedure. In contrast, Cucchetti et al.15 showed a lower early recurrence rate for patients with unfavorable tumor features after AR; while Ishii et al.16 showed a statistically significant difference in overall patient survival, but not in disease-free survival, after AR.
In this study, there were no significant disease-free or overall survival differences between the 2 groups. A tu- mor >5 cm and the presence of cirrhosis were statisti- cally significant risk factors for a recurrence, irrespective of the extent of resection. Considering the tumor size, we concur with Eguchi et al.13 who recommended AR for pa- tients with HCCs sized 2-5 cm to improve survival. In general, HCCs sized <2 cm show a low prevalence of micrometastases and more favorable tumor differentiation.
Conversely, tumors >5 cm have more aggressive biology.
Both situations might diminish the oncologic benefits of AR. Unfortunately, we could not stratify our patients ac- cording to the size of the HCC because of the small-sized
cohort.
Intrahepatic metastases or multicenteric occurrence are 2 common explanations for intrahepatic recurrence of HCC after liver resection. Intrahepatic metastasis can oc- cur in 2 ways involving either locoregional direct spread of tumor cells or systemic intrahepatic metastases by cir- culating tumor cells.17 When tumors recur in multiple cen- ters or by systemic intrahepatic metastases, they cannot be cured surgically by increasing the extent of liver resection. Accordingly, this should be attempted if locore- gional direct dissemination of tumor cells is the cause of intrahepatic recurrence, and if performing AR could pre- vent it. However, in the present study only 1 patient showed an early marginal recurrence after NR, implying that a locoregional direct spread is not the main route of intrahepatic recurrence in patients with a solitary HCC without macroscopic vascular invasion. Marubashi et al.18 reported very similar results to our study, and further in- dicated that NR performed with a surgical margin of 5-10 mm leads to negligible recurrence by local dissemination for both AR and NR.
NR has a benefit in managing patients with HCC. First, by preserving the liver parenchyma as much as possible, postoperative liver function is better than AR, as indicated by our results. Second, the chance of performing multi- modality treatment is higher on tumor recurrence, and re- peat resections can be performed. Third, when salvage liv- er transplantation is considered, a prior AR might lead to difficulties in dissecting the hilum and severe adhesions, resulting in massive bleeding.19
The present study had some limitations that must be taken into consideration. The small-scale, retrospective, observational nature of the study might have influenced the results, and some selection bias was inevitable.
Another limitation is that we did not analyze the gross classification of the tumors, which is a potential risk fac- tor for recurrence.20 Therefore, a large-scale prospective randomized trial would be ideal for investigating surgical outcomes according to the extent of hepatic resection.
In conclusion, the operative outcomes and the patterns of recurrence were similar between the AR and NR groups for treatment of a solitary HCC without macro- scopic vascular invasion. In particular, marginal recur- rences were negligible in both groups. Therefore, we sug- gested that NR could be a good alternative treatment op-
tion for patients with a solitary HCC by balancing the need to maintain an adequate hepatic reservoir with onco- logic clearance. However, further studies are required to confirm the effects of the extent of hepatic resection in such cases.
REFERENCES
1. El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology 2007;132:2557-2576.
2. Tung-Ping Poon R, Fan ST, Wong J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg 2000;232:10-24.
3. Toyosaka A, Okamoto E, Mitsunobu M, Oriyama T, Nakao N, Miura K. Intrahepatic metastases in hepatocellular carcinoma:
evidence for spread via the portal vein as an efferent vessel. Am J Gastroenterol 1996;91:1610-1615.
4. Kaibori M, Matsui Y, Hijikawa T, Uchida Y, Kwon AH, Kamiyama Y. Comparison of limited and anatomic hepatic re- section for hepatocellular carcinoma with hepatitis C. Surgery 2006;139:385-394.
5. Tanaka K, Shimada H, Matsumoto C, Matsuo K, Nagano Y, Endo I, et al. Anatomic versus limited nonanatomic resection for solitary hepatocellular carcinoma. Surgery 2008;143:607-615.
6. Poon RT. Differentiating early and late recurrences after re- section of HCC in cirrhotic patients: implications on surveil- lance, prevention, and treatment strategies. Ann Surg Oncol 2009;16:792-794.
7. Chun JM, Kwon HJ, Sohn J, Kim SG, Park JY, Bae HI, et al.
Prognostic factors after early recurrence in patients who under- went curative resection for hepatocellular carcinoma. J Surg Oncol 2011;103:148-151.
8. Poon RT, Fan ST, Ng IO, Wong J. Significance of resection margin in hepatectomy for hepatocellular carcinoma: a critical reappraisal. Ann Surg 2000;231:544-551.
9. Shoup M, Gonen M, D'Angelica M, Jarnagin WR, DeMatteo RP, Schwartz LH, et al. Volumetric analysis predicts hepatic dys- function in patients undergoing major liver resection. J
Gastrointest Surg 2003;7:325-330.
10. Guglielmi A, Ruzzenente A, Conci S, Valdegamberi A, Iacono C. How much remnant is enough in liver resection? Dig Surg 2012;29:6-17.
11. Suh KS. Systematic hepatectomy for small hepatocellular carci- noma in Korea. J Hepatobiliary Pancreat Surg 2005;12:365-370.
12. Hasegawa K, Kokudo N, Imamura H, Matsuyama Y, Aoki T, Minagawa M, et al. Prognostic impact of anatomic resection for hepatocellular carcinoma. Ann Surg 2005;242:252-259.
13. Eguchi S, Kanematsu T, Arii S, Okazaki M, Okita K, Omata M, et al; Liver Cancer Study Group of Japan. Comparison of the outcomes between an anatomical subsegmentectomy and a non-anatomical minor hepatectomy for single hepatocellular car- cinomas based on a Japanese nationwide survey. Surgery 2008;
143:469-475.
14. Okamura Y, Ito T, Sugiura T, Mori K, Uesaka K. Anatomic ver- sus nonanatomic hepatectomy for a solitary hepatocellular carci- noma: a case-controlled study with propensity score matching.
J Gastrointest Surg 2014;18:1994-2002.
15. Cucchetti A, Qiao GL, Cescon M, Li J, Xia Y, Ercolani G, et al. Anatomic versus nonanatomic resection in cirrhotic patients with early hepatocellular carcinoma. Surgery 2014;155:512-521.
16. Ishii M, Mizuguchi T, Kawamoto M, Meguro M, Ota S, Nishidate T, et al. Propensity score analysis demonstrated the prognostic advantage of anatomical liver resection in hepatocellular carcinoma. World J Gastroenterol 2014;20:3335-3342.
17. Sakon M, Nagano H, Nakamori S, Dono K, Umeshita K, Murakami T, et al. Intrahepatic recurrences of hepatocellular carcinoma after hepatectomy: analysis based on tumor hemodynamics. Arch Surg 2002;137:94-99.
18. Marubashi S, Gotoh K, Akita H, Takahashi H, Sugimura K, Miyoshi N, et al. Analysis of recurrence patterns after anatomical or non-anatomical resection for hepatocellular carcinoma. Ann Surg Oncol 2015;22:2243-2252.
19. Lee SG. Salvage living-donor liver transplantation to previously hepatectomized hepatocellular carcinoma patients: is it a reason- able strategy? Hepatobiliary Pancreat Dis Int 2013;12:10-11.
20. Hui AM, Takayama T, Sano K, Kubota K, Akahane M, Ohtomo K, et al. Predictive value of gross classification of hepatocellular carcinoma on recurrence and survival after hepatectomy. J Hepatol 2000;33:975-979.