BRCA1-associated protein 1 expression and prognostic role in prostate adenocarcinoma
Harim Oh1 , Yoo Jin Lee1 , Sung Gu Kang2 , Bokyung Ahn1 , Eojin Kim1 , Yang-Seok Chae1 , Youngseok Lee1 , Jeong Hyeon Lee1 , Chul Hwan Kim1
Departments of 1Pathology and 2Urology, Korea University Anam Hospital, Korea University College of Medicine, Seoul, Korea
Purpose: As prostate cancer (PCa) is the second most commonly diagnosed cancer worldwide, finding novel markers for prognosis is crucial. BRCA1-associated protein 1 (BAP-1), a nuclear-localized deubiquitinating enzyme, has been reported in several human cancers. However, its prognostic role in PCa remains unknown. Herein, we assessed the prognostic and clinicopathologic signifi- cance of BAP-1 in PCa.
Materials and Methods: Seventy surgical specimens from radical prostatectomy cases were examined. Two cores per case were selected for construction of tissue microarrays (TMAs). After the exclusion of two cases because of tissue sparsity, BAP-1 immuno- histochemical expression was evaluated in 68 cases of formalin-fixed, paraffin-embedded TMA tissue blocks. The immunohisto- chemical stain was scored according to proportion of nuclear staining: negative (<10% of tumor cells) or positive (≥10% of tumor cells).
Results: BAP-1 expression was negative in 30 cases (44.1%) and positive in 38 cases (55.9%). Positive BAP-1 expression was more common in pT3b disease than in pT2 (p=0.038). A high preoperative prostate-specific antigen level was correlated with BAP-1 ex- pression (p=0.014). Age, lymphovascular invasion, perineural invasion, and grade group were not significantly correlated with BAP- 1 expression. Patients with positive BAP-1 expression showed significantly shorter disease-free survival (p=0.013). Additionally, BAP-1 was an independent prognostic factor of PCa (p=0.035; hazard ratio, 9.277; 95% confidence interval, 1.165–73.892).
Conclusions: Our study findings showed an association of BAP-1 expression with poor PCa prognosis and suggest a potential role for BAP-1 as a prognostic biomarker for PCa.
Keywords: BAP1 protein; Prognosis; Prostatic neoplasms; Survival
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: September 17, 2019 • Accepted: November 2, 2019
Corresponding Author: Chul Hwan Kim https://orcid.org/0000-0003-2026-8824
Department of Pathology, Korea University Anam Hospital, Korea University College of Medicine, 73 Goryeodae-ro, Seongbuk-gu, Seoul 02841, Korea TEL: +82-2-920-5590, FAX: +82-2-920-6576, E-mail: [email protected]
ⓒ The Korean Urological Association
www.icurology.org
Investig Clin Urol 2020;61:166-172.
https://doi.org/10.4111/icu.2020.61.2.166 pISSN 2466-0493 • eISSN 2466-054X
INTRODUCTION
Prostate cancer (PCa) is diagnosed in 13.5% of male cancer patients worldwide, making it the second most fre- quently diagnosed cancer in men [1]. PCa usually appears in a localized form, but clinical outcomes vary considerably.
Current clinical prognostic factors are very limited in their ability to improve individual triaging of patients to refine prognosis or recurrent risk. Therefore, it is crucial to inves- tigate PCa progression factors and to find novel biomarkers for predicting PCa prognosis.
BRCA1-associated protein 1 (BAP-1), a deubiquitinating
protein [2], regulates cell cycles and DNA damage by interac- ting with the BRCA1/BARD1 tumor-suppressor heterodimer [3]. Also, nuclear-localized BAP-1 functions as an independent inhibitor in cell proliferation and as a regulator of apoptosis [4]. In addition to these growth inhibitory functions, BAP- 1 also acts as an activator of cell proliferation. By forming complexes with host cell factor 1 (HCF-1) and through deu- biquitination, BAP-1 acts as a cell cycle progression factor at the G1/S transition [5]. Depletion of BAP-1 has even been shown to slow the S phase [3]. Thus, BAP-1 plays dual roles in cell cycle regulation as an activator and deactivator [6].
In recent years, a BAP-1 loss-of-function mutation has been described for several cancers. According to earlier re- ports, a germline BAP-1 mutation is associated with several hereditary cancer syndromes, including uveal melanoma, malignant pleural mesothelioma, clear cell renal cell carcino- ma, and cholangiocarcinoma [7-10]. Additional studies identi- fied somatic BAP-1 mutations that were also associated with corresponding cases [11-13]. However, the view of BAP-1 as a prognostic biomarker is controversial. Patients with BAP- 1 loss-of-function mutations have shorter overall survival in clear cell renal cell carcinoma and uveal melanoma [14]. In contrast, malignant pleural mesothelioma patients with high BAP-1 expression have shorter overall survival [15]. Thus, the prognostic role of BAP-1 depends on cancer type.
A previous study showed that immunohistochemical staining for BAP-1 protein was a good surrogate marker for locating BAP-1 mutations [16]. Thus, BAP-1 protein analysis by immunohistochemistry (IHC) might be useful for prog- nostic evaluation during routine work-up. Here, we evalu- ated BAP-1 expression in PCa and estimated the correlations between BAP-1 and clinicopathologic parameters, as well as disease-free survival.
MATERIALS AND METHODS
1. Patients and samples
From January 2009 to November 2013, surgical speci- mens from patients diagnosed with prostate adenocarcinoma who underwent radical prostatectomy at Korea University Anam Hospital were examined. Patients who had other primary malignancy were excluded. Finally, 70 cases were chosen. None of the patients received neoadjuvant chemo- therapy. Patients’ clinicopathologic features, including age at diagnosis, preoperative prostate-specific antigen (PSA) level, lymphovascular invasion, perineural invasion, metastasis status, pathologic T-stage, and disease-free survival, were retrospectively obtained. One pathologist (YJL) reevaluated the tumors and classified them according to recently up-
dated Gleason score and grade group [17]. Pathologic T-stage was modified according to the American Joint Committee on Cancer (AJCC) 8th edition, in which all organ-confined PCa cases were assigned as pT2 without subclassification [18]. The cutoff point for biochemical recurrence was per- sistent PSA >0.2 ng/mL [19]. The present study protocol was reviewed and approved by the Institutional Review Board of Korea University College of Medicine (approval number:
2019AN0125).
2. Tissue microarray
Two cores were obtained from each formalin-fixed, paraffin-embedded prostate adenocarcinoma tissue block.
The 3-mm-diameter PCa tissue cores were transferred and embedded into a tissue microarray (TMA) block. There were two selection criteria for cores: 1) an area with the highest Gleason score and 2) an area with sufficient tumor extent that was larger than 3 mm in diameter.
3. Immunohistochemical staining
The BAP-1 antibody (clone C-4, 1:50 dilution; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) was used for IHC staining. Sections 4-μm thick from the TMA tissue blocks were stained by use of an automated staining facility (Leica BOND-MAX; Leica Microsystems, Melbourne, Australia).
Normal pancreatic tissue and malignant mesothelioma sec- tions were used as positive and negative controls, respec- tively, for BAP-1 immunostaining.
4. Evaluation of immunohistochemical staining
IHC staining for BAP-1 was interpreted according to the proportion of stained cells. Only nuclear staining was consid- ered, and cytoplasmic staining was regarded as negative. The proportion was scored based on the average staining propor- tion of the two cores. BAP-1 staining was considered positive if any intensity of nuclear staining was noted in ≥10% of tumor cells and negative if staining was noted in <10% of tumor cells (Fig. 1). The cutoff value was set according to the criteria defined previously by Shah et al. [20].5. Statistical analysis
All statistical analyses were performed using PASW Statistics version 18.0 (SPSS Inc., Chicago, IL, USA). The cor- relation between BAP-1 expression and clinicopathologic fea- tures of prostate adenocarcinoma were calculated using uni- variate and multivariate logistic regression analysis. Disease- free survival was analyzed using the Kaplan–Meier method, and the significance of the difference between BAP-1 ex- pression was analyzed using the log-rank test. Each clinical
factor was evaluated for independent prognostic significance using the Cox proportional-hazards regression model, and factors yielding a p-value <0.05 in univariate analyses were included in the multivariate model. A p-value <0.05 was con- sidered statistically significant.
RESULTS
1. Cohort characterization
Sixty-eight of 70 cases were analyzed for this study. Two cases were excluded because they lacked tumor cells in the TMA. Patient age ranged from 49 to 76 years, with a mean age of 63 years. PSA level ranged from 3.34 to 45.91 ng/mL, with a mean of 11.36 ng/mL. There were 17, 45, 4, and 2 cases with Gleason scores of 6, 7, 8, and 9, respectively. There were 17, 36, 9, 4, and 2 cases in grade group 1 (Gleason score ≤6), group 2 (Gleason score 3+4), group 3 (Gleason score 4+3), group 4 (Gleason score 8), and group 5 (Gleason score 9 or 10), respectively. Lymphovascular invasion was present in 4 cas- es, and perineural invasion was present in 35 cases. Among the 68 cases, 47, 10, and 11 cases were stage pT2, pT3a, and pT3b, respectively. All cases were of localized PCa without metastasis at the time of diagnosis. The median follow-up time was 92 months (range, 2–120 months).
2. Relationships between BAP-1 and clinicopatho- logic features
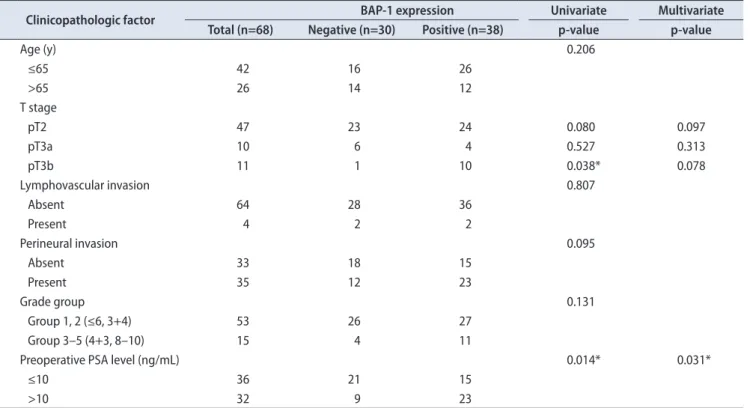
Of the 68 cases, BAP-1 expression was negative in 30 (44.1%) and positive in 38 (55.9%). BAP-1-positive PCa was significantly correlated with higher preoperative PSA level by univariate logistic regression analysis (p=0.014; odd ratio, 3.578). BAP-1 expression was more common in pT3b disease than in pT2 (p=0.038; odd ratio, 9.583). Age, grade group, lym- phovascular invasion, and perineural invasion showed no significant correlation with BAP-1 expression. By multivari- ate logistic regression analysis, preoperative PSA level was significantly correlated with BAP-1 expression (p=0.031; odd ratio, 3.350; Table 1).
3. Correlation of BAP-1 expression with disease- free survival of prostate adenocarcinoma pa- tients
Ten of the 68 total cases experienced biochemical recur- rence: 1 had negative BAP-1 expression, and the other 9 cases had positive BAP-1 expression. Because survival was greater than 50% at the last time point, median survival was unde- fined. The 86-month survival percentages of BAP-1-negative and BAP-1-positive cases were 96.6% and 72%, respectively.
Patients with positive BAP-1 expression had significantly shorter disease-free survival (p=0.013; Fig. 2). There was no cancer-related death in the 68 cases.
Univariate survival analysis via the Cox proportional- hazards regression model showed that BAP-1 expression was related to shorter disease-free survival (p=0.040). Pre- operative PSA level (>10 ng/mL), higher grade group, and lymphovascular invasion were significantly associated with shorter disease-free survival (p=0.025, 0.003, and 0.001, respec- tively). T-stage, perineural invasion, resection-margin status, and age were not significantly correlated with disease-free survival. In the multivariate survival analysis including preoperative PSA level, grade group, and lymphovascular invasion by back variable selection using the Wald method, BAP-1 expression and lymphovascular invasion were final independent variables that were significantly correlated with disease-free survival (p=0.035 and 0.001, respectively;
Table 2).
DISCUSSION
Ubiquitin attaches to proteins and causes post-transla- tional modification that either alters target protein function or suppresses protein expression. Deubiquitinase removes ubiquitin via hydrolysis. Depending on its target, the func- tion of deubiquitinase on cellular regulation could differ.
When tumor suppressors are deubiquitinated, their expres- sion is preserved, preventing uncontrolled cellular prolifera- tion. In this case, deubiquitinase acts as a tumor suppressor.
A B
Fig. 1. Immunohistochemical staining for BRCA1-associated protein 1 (BAP-1):
(A) BAP-1 negative, (B) BAP-1 positive (all cases 400×).
Overall, the role of BAP-1, a nuclear-localized deubiquitinat- ing protein [2], could differ according to target protein.
Earlier studies reported that BAP-1 binds to the RING finger motif of BRCA1 and enhances the BRCA1-mediated cellular growth suppressor function by deubiquitination [2].
By serving as a regulator of the BRCA1 pathway, BAP-1 acts as a tumor suppressor.
However, several other studies reported that BAP-1 participates in cell proliferation. BAP-1 depletion by RNAi inhibits proliferation of breast cancer cells (MCF-7 and T-
47D). Furthermore, previous research argued that the interaction between BAP-1 and HCF-1 is critical for BAP- 1-mediated cell growth. HCF-1, a cell cycle modulator, is a heterodimer composed of HCF-1N and HCF-1C. In particular, BAP-1 regulates cell proliferation by deubiquitinating HCF- 1N, which promotes G1-phase progression and S-phase entry [3,21]. Another study revealed that BAP-1 loss downregulates the downstream targets of E2F, an important cell cycle pro- gression factor, including cyclin A2, E2F1, p107, and CDC25C [11]. Taken together, these findings indicate that BAP-1 may play a dual role in cellular growth, both preventing exces- sive growth and ensuring appropriate cell growth.
BAP-1 status can be evaluated by three methods. First, genomic hybridization can inspect for deletions in the BAP-1 gene locus at 3p21. Second, the BAP-1 gene can be sequenced to identify any mutations. Third, IHC can detect BAP-1 pro- teins [20]. Evaluating BAP-1 protein expression via IHC is considered the superior method for evaluating BAP-1’s func- tional status, rather than evaluating deletions or mutations in coding lesions [16]. Moreover, because most BAP-1 muta- tions cause rapid degradation of proteins or reduce nuclear localization, BAP-1 mutations are highly associated with negative immunohistochemical staining [22].
To our knowledge, this is the first study to provide evi- dence of correlations between BAP-1 expression and clinico- Table 1. The correlation between BAP-1 expression and clinicopathologic factors
Clinicopathologic factor BAP-1 expression Univariate Multivariate
Total (n=68) Negative (n=30) Positive (n=38) p-value p-value
Age (y) 0.206
≤65 42 16 26
>65 26 14 12
T stage
pT2 47 23 24 0.080 0.097
pT3a 10 6 4 0.527 0.313
pT3b 11 1 10 0.038* 0.078
Lymphovascular invasion 0.807
Absent 64 28 36
Present 4 2 2
Perineural invasion 0.095
Absent 33 18 15
Present 35 12 23
Grade group 0.131
Group 1, 2 (≤6, 3+4) 53 26 27
Group 3–5 (4+3, 8–10) 15 4 11
Preoperative PSA level (ng/mL) 0.014* 0.031*
≤10 36 21 15
>10 32 9 23
BAP-1, BRCA1-associated protein 1; PSA, prostate-specific antigen.
*p<0.05.
0 20 40 60 80 100
1.0
0.8
0.6
0.4
0.2
120
Survivalprobability
Disease free survival (mo) 0
BAP-1 negative (n=30)
BAP-1 positive (n=38) p=0.013 + + + + + + + + + + + ++ + ++
++ + + +
+
+ +
+ +++ ++ + + +++ ++++ ++ ++ +
Fig. 2. Kaplan–Meier curves showing association of disease-free sur- vival with BRCA1-associated protein 1 (BAP-1) expression.
pathologic factors in PCa cases using IHC. In our study, PCa cases with positive BAP-1 expression had significantly high- er T-stage and higher PSA level. Consistently, PCa patients with positive BAP-1 expression had shorter disease-free survival than did patients with negative BAP-1 expression.
Also, high preoperative PSA level, high grade group, and lymphovascular invasion were significantly associated with shorter disease-free survival. However, a preexisting staging score, including T-stage, extraprostatic extension, and semi- nal vesicle invasion, were not correlated with disease-free survival. This result could be explained by the relatively small number of cases. Despite our limited sample size, our study adds to the literature suggesting BAP-1 immunoex- pression as a potential prognostic biomarker.
Although BAP-1 has been discussed in a broad spectrum of cancers, consensus has not been reached for it prognostic role. BAP-1 loss is associated with poor prognosis in clear cell renal cell carcinoma and uveal melanoma [14]. In contrast, however, BAP-1 loss was found to have a protective role in malignant mesothelioma, which is consistent with our re- sults for PCa [15]. These conflicting results support the view that BAP-1 may have many yet unknown biological func- tions, and that its activity might have a tissue-specific pat- tern. Especially in PCa cases, the BAP-1 gene may play an oncogenic role rather than a generally well-known tumor- suppressor role.
Since BAP-1 functions as a deubiquitinating enzyme, it can act as either an oncogene or a tumor-suppressor gene, depending on the target protein for deubiquitination. As previously mentioned, cell cycle proliferation pathways such as the HCF-1 molecular pathway and E2F related proteins
would be affected by BAP-1 oncogenic action [3,11]. Other molecular pathways associated with BAP-1 need to be clari- fied in further studies.
BAP-1 was initially discovered as a BRCA1-associated protein [2], and many studies are now attempting to identify the association between BAP-1 and BRCA1, especially the pathways associated with histone H2A, heterochromatin, and DNA damage response [23]. A study by Omari et al.
[24] reported that BRCA1 gene loss along with BRCA1 gene gain is associated with ALDH1 expression, which is a stem cell marker. This indicates that BRCA1 gene gain is associ- ated with PCa dedifferentiation. The poor prognosis among patients with positive BAP-1 expression might be explained by BRCA1 gene gain affecting dedifferentiation. Also, the cooperative activity between BRCA1 and BAP-1 could be a future drug target.
Our study limitations included our relatively small cohort study size and the lack of genomic or mutational testing in our analyses. Despite these limitations, ours is the first study to correlate BAP-1 immunoexpression with clinicopathologic factors in PCa, and we revealed that BAP- 1 is a potential prognostic marker for PCa. Larger-scaled and molecular studies will further enhance the validity of our research, and they could also serve to elucidate new thera- peutic strategies. Finally, IHC for BAP-1 may be used for preoperative biopsy specimens for PCa treatment planning.
CONCLUSIONS
The purpose of our study was to determine whether BAP-1 IHC can stratify PCa prognoses in a clinically useful Table 2. Cox proportional hazard regression model survival analysis
Clinicopathologic factors Univariate analysis Multivariate analysis
HR 95% CI p-value HR 95% CI p-value
T stage
pT2 1 0.338
pT3a 0.917 0.110–7.623 0.936
pT3b 2.773 0.676–11.365 0.157
Lymphovascular invasion, absent vs. present 11.007 2.725–44.452 0.001* 11.789 2.838–48.969 0.001*
Perineural invasion, absent vs. present 4.178 0.884–19.752 0.071 Grade, group 1,2 vs. group 3–5 6.558 1.862–23.103 0.003*
Extraprostatic extension, absent vs. present 1.826 0.511–6.530 0.354 PSA level (ng/mL), ≤10 vs. >10 5.938 1.255–28.099 0.025*
Seminal vesicle invasion, absent vs. present 2.809 0.709–11.128 0.141
Age (y), ≤65 vs. >65 0.890 0.251–3.161 0.857
Resection-margin status, negative vs. positive 1.111 0.313–3.950 0.871
BAP-1 expression, negative vs. positive 8.734 1.104–69.096 0.040* 9.277 1.165–73.892 0.035*
HR, hazard ratio; CI, confidence interval; BAP-1, BRCA1-associated protein 1; PSA, prostate-specific antigen.
*p<0.05.
way. In our dataset, BAP-1 immunoexpression predicted poor prognosis in PCa patients. Analyzing BAP-1 expression by IHC may be a valuable way to stratify more aggressive PCa cases from less aggressive cases. The precise role of BAP-1 in PCa deserves further investigation and validation in future studies.
CONFLICTS OF INTEREST
The authors have nothing to disclose.
AUTHORS’ CONTRIBUTIONS
Research conception and design: Yoo Jin Lee and Yang- Seok Chae. Data acquisition: Sung Gu Kang, Bokyung Ahn, and Eojin Kim, Statistical analysis: Harim Oh and Young- seok Lee. Data analysis and interpretation: Harim Oh, Yoo Jin Lee, Jeong Hyeon Lee, and Chul Hwan Kim. Drafting of the manuscript: Harim Oh. Critical revision of the manu- script: Chul Hwan Kim. Supervision: Chul Hwan Kim. Ap- proval of the final manuscript: Chul Hwan Kim.
REFERENCES
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A.
Global cancer statistics 2018: GLOBOCAN estimates of inci- dence and mortality worldwide for 36 cancers in 185 countries.
CA Cancer J Clin 2018;68:394-424.
2. Jensen DE, Proctor M, Marquis ST, Gardner HP, Ha SI, Cho- dosh LA, et al. BAP1: a novel ubiquitin hydrolase which binds to the BRCA1 RING finger and enhances BRCA1-mediated cell growth suppression. Oncogene 1998;16:1097-112.
3. Nishikawa H, Wu W, Koike A, Kojima R, Gomi H, Fukuda M, et al. BRCA1-associated protein 1 interferes with BRCA1/
BARD1 RING heterodimer activity. Cancer Res 2009;69:111-9.
4. Ventii KH, Devi NS, Friedrich KL, Chernova TA, Tighiouart M, Van Meir EG, et al. BRCA1-associated protein-1 is a tumor suppressor that requires deubiquitinating activity and nuclear localization. Cancer Res 2008;68:6953-62.
5. Machida YJ, Machida Y, Vashisht AA, Wohlschlegel JA, Dutta A. The deubiquitinating enzyme BAP1 regulates cell growth via interaction with HCF-1. J Biol Chem 2009;284:34179-88.
6. Rai K, Pilarski R, Cebulla CM, Abdel-Rahman MH. Compre- hensive review of BAP1 tumor predisposition syndrome with report of two new cases. Clin Genet 2016;89:285-94.
7. Testa JR, Cheung M, Pei J, Below JE, Tan Y, Sementino E, et al. Germline BAP1 mutations predispose to malignant esothe- lioma. Nat Genet 2011;43:1022-5.
8. Popova T, Hebert L, Jacquemin V, Gad S, Caux-Moncoutier V,
Dubois-d'Enghien C, et al. Germline BAP1 mutations predis- pose to renal cell carcinomas. Am J Hum Genet 2013;92:974- 80.
9. Wiesner T, Obenauf AC, Murali R, Fried I, Griewank KG, Ulz P, et al. Germline mutations in BAP1 predispose to melanocytic tumors. Nat Genet 2011;43:1018-21.
10. Klebe S, Driml J, Nasu M, Pastorino S, Zangiabadi A, Hender- son D, et al. BAP1 hereditary cancer predisposition syndrome:
a case report and review of literature. Biomark Res 2015;3:14.
11. Bott M, Brevet M, Taylor BS, Shimizu S, Ito T, Wang L, et al.
The nuclear deubiquitinase BAP1 is commonly inactivated by somatic mutations and 3p21.1 losses in malignant pleural me- sothelioma. Nat Genet 2011;43:668-72.
12. Harbour JW, Onken MD, Roberson ED, Duan S, Cao L, Wor- ley LA, et al. Frequent mutation of BAP1 in metastasizing uveal melanomas. Science 2010;330:1410-3.
13. Peña-Llopis S, Vega-Rubín-de-Celis S, Liao A, Leng N, Pavía- Jiménez A, Wang S, et al. BAP1 loss defines a new class of renal cell carcinoma. Nat Genet 2012;44:751-9.
14. Wang XY, Wang Z, Huang JB, Ren XD, Ye D, Zhu WW, et al.
Tissue-specific significance of BAP1 gene mutation in prog- nostic prediction and molecular taxonomy among different types of cancer. Tumour Biol 2017;39:1010428317699111.
15. Arzt L, Quehenberger F, Halbwedl I, Mairinger T, Popper HH.
BAP1 protein is a progression factor in malignant pleural me- sothelioma. Pathol Oncol Res 2014;20:145-51.
16. Wiesner T, Murali R, Fried I, Cerroni L, Busam K, Kutzner H, et al. A distinct subset of atypical Spitz tumors is characterized by BRAF mutation and loss of BAP1 expression. Am J Surg Pathol 2012;36:818-30.
17. Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Hum- phrey PA; Grading Committee. The 2014 International Society of Urological Pathology (ISUP) consensus conference on Glea- son grading of prostatic carcinoma: definition of grading pat- terns and proposal for a new grading system. Am J Surg Pathol 2016;40:244-52.
18. Amin MB; American Joint Committee on Cancer. AJCC can- cer staging manual. 8th ed. New York: Springer; 2017.
19. Freedland SJ, Sutter ME, Dorey F, Aronson WJ. Defining the ideal cutpoint for determining PSA recurrence after radical prostatectomy. Prostate-specific antigen. Urology 2003;61:365- 9.
20. Shah AA, Bourne TD, Murali R. BAP1 protein loss by im- munohistochemistry: a potentially useful tool for prognos- tic prediction in patients with uveal melanoma. Pathology 2013;45:651-6.
21. Julien E, Herr W. Proteolytic processing is necessary to sepa- rate and ensure proper cell growth and cytokinesis functions of HCF-1. EMBO J 2003;22:2360-9.
22. Luchini C, Veronese N, Yachida S, Cheng L, Nottegar A, Stubbs B, et al. Different prognostic roles of tumor suppressor gene BAP1 in cancer: a systematic review with meta-analysis. Genes Chromosomes Cancer 2016;55:741-9.
23. Fukuda T, Tsuruga T, Kuroda T, Nishikawa H, Ohta T. Func- tional link between BRCA1 and BAP1 through histone H2A,
heterochromatin and DNA damage response. Curr Cancer Drug Targets 2016;16:101-9.
24. Omari A, Nastaly P, Balabas A, Dąbrowska M, Bielińska B, Huss S, et al. Somatic aberrations of BRCA1 gene are associ- ated with progressive and stem cell-like phenotype of prostate cancer. bioRxiv 2018:271312.