Journalofthe Korean ChemiculSociety 2000,L’d,44, No 3
Printedin the Republic of Korea
Spectrophotometric Determination of Nickel (II) in lkveen80 Micellar Medium
Seung-Kwon Lee and Hee-Seen Choi*
Depannrent of Chemistry The University of Suwon, Suwon 445-743, Korva (Received December 30, 1999)
$2 Q/. Tween80 u]’$ -$441+ 4%x@-~j APDC% “l-f!++ Ni(IIE ‘j%@k “c!%ql ~~~ %?~}
%+. Ni(PDC), ‘%4 % ~~~?j~ chlorofonM1’j Y-% Tween8&l]~j Lj %+ E“J+ Lj +%+ zJE
% ~~ ~%~. Ni(PDC)2 ‘%+ pH 7.001]~j zXZ 1W@ ‘j~7’}~] uI-?- %~x~~}%-$%, APDC% Ni(II) Ql M+ lmti “1’J %J~ +Q Z!%4= %01 %’d~%~. Tween8M %-~% 0.1%7} 4@%93, 0.1% Tween80 -%qoll~j Ni(PDC), ~%~ %xj+x~~ % ~~~~~ (R2=0.9955)-% ~~~%~. “1 %~~~
Zj+&JZ]~ xj%EJ41+- 4-’+ 0.09 @rnU+ 0.28MATDIWPI ,7&4~o11 +%%! %+5++ 100% ~+
~% al I-F+&+. H]% 4 ~j
~+li%ol Pj-W d-zl ”J ,4zIAlsdl %+9%
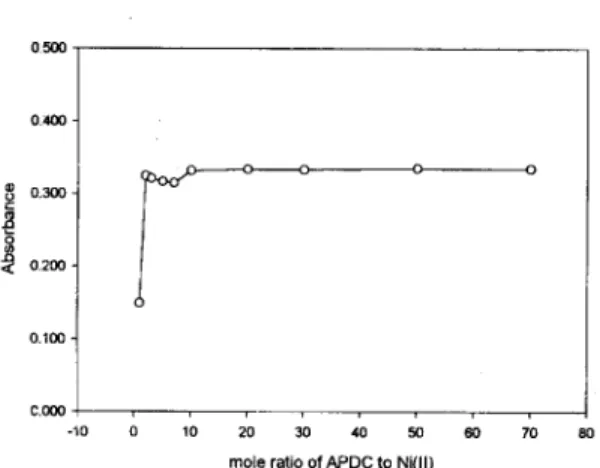
Ni(H )% Zi %+%~1 01 -%9 + 5+% A“lq.ABSTRACT. We have studied on the determination of Ni(H) using APDC as a completing agent in Tween80 miceikr medium. The abso@on spectrum of Ni(PDC), complex in Tween80 medium was better defined and more sensitive than that in chloroform. Ni(PDC), complex was very stable at pH 7.0 and up to 100 minutes, and could be quantitatively chelated when APDC was added to over 10 times moles of Ni(II).
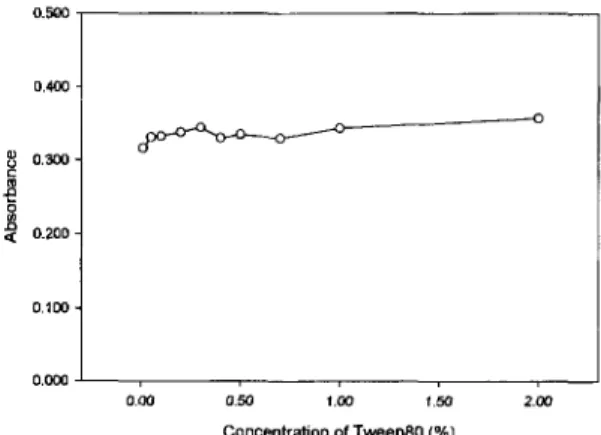
The optimum concentration of Tween80 was 0.1 %. The calibration curve of Ni(PDC)~ complex with good lin- earity(R2=0.9955) was obtained in 0.1YO Tween80 medium. The detection limit and the determination limit were 0.09 ~g/mL and 0.28 pg/mL, respectively. This technique was applied to the analysis of Suwon stream water samples, and about 100% of recoveries were obtained from the spiked samples. Although the formation of Ni(PDC)* complex was interfered by various metal ions, this technique could be applied to the practical determination of Ni(II).
– 207
0.500.
am
g 0.302
ii O.*C9
o.lm
O.m
0 2 4 e 8 10
PH
O,smoI
320 325 350 375 41XI 425 4EQ 475 502
Wavelength (rim)
Fig. 2. Absorption spectra of Ni(PDC)z complex in chloro- form and Tween80 medium,
2000,Vol.44, No. 3
0,4W
g O,wxl
1
$ 0.200
0.100
O.wo
0
0.03 0.52 1,72 1.30 2.CO
Concentration of Tween80 (% I
210
05CW
O&K-
g 0.3W
~
$ 02WJ.
O.lCC-
C.rm 0
-100102030 4050 S07080
moleratioof APDc to Ni(ll)
Fig. 4. Effect of the amount of APDC car the absorbance of Ni(PDC)2 in Tween80 medium.
Table 2.Recoveries of Ni(II)in Suwon stream water
Table 1.Tolerant limits* of various interfering species on the determination of Ni(II)(l @nL)
Tolerant mole ratio Metal ions
103 K+
50 Cr(III)
30 Na+, Mn(II), Zn(II)
10 Al(III), Fe(III), Sn(II)
5 CU(II), Co(II)
1 Bi(III), Cd(II), Pb(II)
Spiked(g/mL) 0.00 0.50 1.00
Measured(g/mI.)*
Recovery(%)
this method IcP/Ms
0.00 0.00
0.51(4.02) 0.50(@.02)
102
1.05(4.04) 1.01(4.03) 105
*Measured values and their standard deviations were obtained using seven samples
1. Kim, Y. S.; Jung, Y. J.; Choi, H. S. Bull. Korean Chem.
SOC. 1998.19, 50.
2. Tao, H.; Miyazaki, A.; Bansho, K.; Umezaki, Y. Anal.
Chim. Acts 1984, 156, 159.
3. Lo, J. M.; Yu, J. C.; Hutchison, F. L; Wai, C. M. Anal.
Chem. 1982, 54, 2536.
4. Diaz Garcia, M. E.; Sanz Medel, A. 7idanta 1986,33, 255.
5. Hayashi, K.; Sasaki, Y.; Tagashira, S.; Kosaka, E. Anal.
Chem. 1986, 58, 1444.
6. Paradkq R. P.; Williams, R. R. AnaL C!-zem.1994,66, 2752.
7. San Andres, M. P.;Marind, M. L.; Vera, S. Analyst 1995, 120, 225.
8. Tagashira, S.; Onoue, K.; Morakami, Y.; Sasaki, Y Bull.
Chem. Sot. Jpn. 1992, 65, 286.
9. Vaidya, B.; Porter, M. D. Anal. Chem. 1997,69,2688.
10. Hausenblesova, Z.; Namkova, I.; Suk, V. Microchem. J.
1981, 26, 262.
11. Uesugi, K.; Miyawaki, M. Microchem. J. 1981, 26, 288.
12. Xi-Wen, H.; Poe, D. Talanta 1981, 28, 419.
13. Callahan, J. H.; Kook, K. D. AnaL Chem. 1982,54,59.
14. Jarosz, M.; Marczenko, Z. Analyst 1984, 109, 35.
15. Becher, P. In Sur-jactant Science Series; Schick, M. J., Ed.; Marcel Dekke~ New York, ~. S. A., 1966; Vol. 1, p 559.
16. Okada, T. Anal. Chem. 1992, 64, 2138.
17. Aspila, K. I.; Sastri, V. S.; Chakrabtii, C. L. Talanta 1969, 16, 1099.
18. Esteve-Romero, J. S.; Monferrer-Pons, L1.;Ramis-Ramos, G.; Garcia-Alvarez-Coque, M. C. Talanta 1995,42,737.
19. Becher, P. In Su~actant Science Series; Schick, M. J., Ed.; Marcel Dekker New York U. S. A., 196@Vol. 1, p 481.
20. Lee, J. S.; Choi, J. M.; Choi, H. S.; Kim, Y. S. Ana- lytical Science and Technology 1995, 8, 321.
21. Everson, R. J.; Parker, H. E. Anal. Chern. 1974,46, 1966.
22. Skoog, D. A.; Holler, F. J.; Nieman, T. A. Principles of Instrumental Analysis, 5tb cd.; Saunders College Pub- lishing: U. S. A., 1998, p 13.
2000,vol. 44, No. 3