http://osj.kr
Active Exchange of Water and Nutrients between Seawater and Shallow
Pore Water in Intertidal Sandflats
Dong-Woon Hwang
1,2, Guebuem Kim
2, and Han Soeb Yang
3*
1Tidal-flat Research Center, National Fisheries Research and Development Institute, Gunsan 573-882, Korea 2
School of Earth & Environmental Sciences/RIO, Seoul National University, Seoul 151-742, Korea 3Department of Oceanography, Pukyong National University, Pusan 608-737, Korea
Received 15 July 2008; Revised 11 September 2008; Accepted 15 December 2008
Abstract− In order to determine the temporal and spatial
variations of nutrient profiles in the shallow pore water columns (upper 30 cm depth) of intertidal sandflats, we measured the salinity and nutrient concentrations in pore water and seawater at various coastal environments along the southern coast of Korea. In the intertidal zone, salinity and nutrient concentrations in pore water showed marked vertical changes with depth, owing to the active exchange between the pore water and overlying seawater, while they are temporally more stable and vertically constant in the sublittoral zone. In some cases, the advective flow of fresh groundwater caused strong vertical gradients of salinity and nutrients in the upper 10 cm depth of surface sediments, indicating the active mixing of the fresher groundwater with overlying seawater. Such upper pore water column profiles clearly signified the temporal fluctuation of lower-salinity and higher-Si seawater intrusion into pore water in an intertidal sandflat near the mouth of an estuary. We also observed a semi-monthly fluctuation of pore water nutrients due to spring-neap tide associated recirculation of seawater through the upper sediments. Our study shows that the exchange of water and nutrients between shallow pore water and overlying seawater is most active in the upper 20 cm layer of intertidal sandflats, due to physical forces such as tides, wave set-up, and density-thermal gradient.
Key words− submarine groundwater discharge, pore water,
nutrients, sandflat Regional index terms, Korea, Ilkwang Bay, Nakdong River Estuary, Naro Island
1. Introduction
The concentrations of nutrients in coastal waters are affected by river runoff, exchange with offshore seawater,
atmospheric deposition, diffusion from sediments, submarine groundwater discharge (SGD), and biotic processes (i.e. N-fixation). During the last decade, a number of SGD studies have been conducted in various coastal environments such as salt marshes, river lagoons, and semi-enclosed bays. Some SGD studies have shown that the inputs of SGD-driven nutrients may be comparable to or even higher than those of river discharge in many coastal areas (Corbett et al. 1999; Krest et al. 2000; Charette et al. 2001; Kelly and Moran 2002; Burnett et al. 2003, 2006, 2007; Garrison et
al. 2003; Hwang et al. 2005; Kim et al. 2005; Boehm et al.
2006; Niencheski et al. 2007; Swarzenski et al. 2007). Thus, submarine discharge of both fresh groundwater and re-circulating seawater is now recognized as an important source for dissolved nutrients in the coastal ocean (Burnett
et al. 2003; Kim et al. 2005).
The submarine inputs of fresh groundwater into the coastal zone are basically controlled by terrestrial hydraulic gradients (Burnett et al. 2003). Re-circulating seawater, on the other hand, is not only driven by hydraulic gradients on land, but also by various marine components such as tides, waves, and density-thermal gradients (Rocha 2000; Burnett
et al. 2006; Robinson et al. 2006, 2007). For example, tides
and waves create a change in the hydraulic gradient in the coastal zone, which may lead to advective pore water flows (Shum and Sundby 1996; Huettel and Webster 2001; Burnett et al. 2003). These flows move along two or three-dimensional flow pathways in coastal sediments, and the solute transport caused by pore water flows has been recognized as one of the important SGD processes.
*Corresponding author. E-mail: hsyang@pknu.ac.kr Article
Recently, Martin et al. (2004) reported that the mixing of pore water and estuarine water by bio-irrigation, wave, and tidal pumping has enhanced SGD at the Banana River Lagoon, Florida. Precht and Huettel (2003) also suggested that shallow-water waves could increase the fluid exchange between sandy sediments and overlying water 50-fold, relative to the exchange by molecular diffusion.
Some recent studies have attempted to reveal the impact of SGD on the distributions of salinity, trace elements, and natural radionuclides (Rn and Ra isotopes) in the pore water of permeable sediments. However, these studies are mostly for the geochemistry of deep pore water for the upper 10 m layer of coastal sediments (Cable et al. 2004; Martin et
al. 2004, 2006; Charette et al. 2005; Charette and
Sholkovitz 2006). Thus, in this study, we focused on the distribution of salinity and nutrients in shallow pore water in the upper 30 cm layer of surface sediments, with high depth resolution, in order to look closely at the ventilation processes of nutrients in the intertidal zone where active wave set-up and recirculation of seawater are taking place. Here, we chose Ilkwang Bay, Nakdong River Estuary, and Naro Island on the southern coast of Korea as study areas, which have different geological and oceanographic settings.
2. Materials and Methods
Study areas
The study area, Ilkwang Beach, is located in the western part of Ilkwang Bay in the southeastern part of Korea (Fig.
1A) and has an area of approximately 5.0 × 104
m2
. Ilkwang Bay is semi-enclosed by a rocky-sandy shore and is
relatively small and shallow, with an area of about 1.6 × 106
m2
and a mean depth of ~5 m. The tide is semi-diurnal, with tidal difference of 1.0 m during spring tides. The investigation at Ilkwang Beach was carried out at two sites on August 29 (Stations I1 and I3) and September 2, 2005 (Stations I2 and I4). One of the sampling sites is above the low-tide sea level (Station I1 and I2; Fig. 2A) and repeatedly experienced emersion and immersion through the tidal cycle. The other sampling site is approximately 40 to 80 cm depth below the low-tide sea level and was constantly immersed (Station I3 and I4; Fig. 2A). The sediment is composed mainly of sand, and the seabed around the sampling site has a slightly steep
slope of 2-10 cm m-1
.
The study area, Dadaepo Beach, is located in the southeastern part of the mouth of the Nakdong River (Fig. 1B), whose runoff is very limited due to an artificial dam (Nakdong River Dam) constructed at its downstream end. The beach
area is approximately 1.0 × 105
m2
. The tide in this region is
Fig. 1. A map showing the sampling sites in the intertidal sandflats on (A) Ilkwang Bay on August 29 and September 2, 2005, (B) Nakdong
River Estuary (Dadaepo Beach) from July 29 to August 24, 2003, and (C) Naro Island from July 11 to August 2, 2002, in the southern coast of Korea.
semi-diurnal, with approximately 1.4 m of average tidal difference. The investigation at Dadaepo intertidal sandflat was carried out at one fixed site (Station D2) in August 2003 at two or four day intervals. Since the sampling site is near the low-tide sea level, it experienced emersion and immersion over the tidal cycle (Fig. 2A). In addition, the sampling site is under the influence of wave-setup and outflows from the Dadaepo watershed. The sediment is composed mainly of sand, and the seabed around the
sampling site has a very gentle slope of 0.9 cm m-1
. The sampling area, Naro Island Beach, is one of the beaches connected with the open sea, and is located in the northern part of Wae Naro Island (Fig. 1C). The length and width of this beach is approximately 1,000 m and 100 m
(beach area : 1.0 × 105
m2
), respectively. The tide in this region is semi-diurnal, with approximately 3 m of average tidal difference. The investigation at Naro Island intertidal sandflat was carried out at one sampling site (Station N1) in July 2002 at a one week interval in order to look at spring-neap tide influences. The sampling site is located in the center of the beach and is 30 cm above the low-tide sea level, experiencing emersion and immersion depending on the tidal stage (Fig. 2A). The sediment is composed mainly of sand, and the seabed around the sampling site has a very
gentle slope of 1.9 cm m-1
.
Sampling and analytical methods
Pore water and seawater samples were taken in order to measure salinity and nutrients. During low tide, the samplers for collecting the pore water were inserted directly into the sand sediment by hand pressing. The multi-level samplers were installed in the upper intertidal (stations I1), lower intertidal (stations I2 and D2) and sublittoral (stations I3 and I4) zones of Ilkwang and Dadaepo Beaches, whereas a one-level sampler was installed in the lower intertidal zone (station N1) of Naro Island Beach. The multi-level sampler (Fig. 2B) is made up of nine to ten polypropylene tubes (2 mm inner and 3 mm outer diameter) at intervals of a few cm, tied to a stainless steel plate (7 cm width and 50 cm length). The dead volume of the tubes in this multi-level sampler was less than approximately 1.5 mL. In order to avoid the cross-contamination of samples from different sediment depths, the polypropylene tubes were fixed with silicone gel and then wrapped with insulation tape, leaving only their tips with the sampling ports exposed.
At the Ilkwang intertidal sandflat, pore water samples were collected from the upper 40 cm sediments at intervals of 2 or 5 cm during a series of half tidal cycle (high to low tide, low tide, low to high tide) on August 29 and September 2, 2005. Hence, the pore water data in this study area do not represent synchronous temporal and spatial distributions of the measured parameters. At the Dadaepo intertidal sandflat, pore water samples were collected from the upper 30 cm sediments at an interval of approximately 3 cm at the low tide from July 29 to August 24, 2003. At the Naro Island
Fig. 2. A diagram showing (A) the sampling location at the
intertidal and sublittoral sandflat and (B) the design of a multi-level sampler used throughout this study in order to collect shallow pore water from coastal sediments. Pore water samples were collected at intervals of 2 or 5 cm continuously over a half tidal cycles for Ilkwang Bay, at an interval of 3 cm at low tide for the Nakdong River Estuary, and at 5 cm depth below the sediment surface at low tide for Naro Island.
intertidal sandflat, pore water samples were collected at approximately 5 cm below the surface sediment at the low tide from July 11 to August 2, 2002. Here, the pore water samples in the intertidal region were taken either immediately before or after submergence.
About 7~8 mL of the pore water samples were obtained slowly with the multi-level and one-level samplers, which is coupled to a 10 mL vacuum tube and a polyethylene syringe, respectively. The pore water samples were collected simultaneously at all depths. In addition, the pore water samples in the intertidal zone were collected within 15 minute after emersion. The overlying seawater samples were collected using a plastic bucket.
Temperature and salinity were measured in the field using a portable salinometer (WTW, Model LF340 and ISTEK, Model 47C) immediately after the water samples were
collected. Nutrient (NO3
− , NO2 − , NH4 + , PO4 3− , and Si(OH)4) samples were collected in vacuum tubes (~10 mL) or polyethylene bottles (~50 mL), and frozen until analyses. Nutrients were analyzed using an Auto-Analyzer (Bran+ Lubbe K.K, Model TRAACS 2000 and Alliance, Model Futura) in the laboratory. The detection limit for nutrients
using the Auto-Analyzer was about 0.03 μM. Most of
nutrient experiments were performed in duplicate using the same method. In this study, we define DIN as the sum of
NO3 − , NO2 − , and NH4 + , DIP as PO4 3−
, and DSi as Si(OH)4.
3. Results and Discussion
High-low tide variations in the Ilkwang embayment
Although the pore water and seawater samples from the Ilkwang intertidal sandflat were not collected simultaneously as mentioned above, salinity and nutrient concentrations in surface seawater at stations I3 and I4 did not show large spatial variation and were constant during the sampling times (within ~ 6 hours). However, salinity and nutrient concentrations in the pore water of surface sediment showed interesting temporal and spatial distributions (Fig. 3). Firstly, salinity of the pore water at stations I1 and I4 was relatively uniform with depth, whereas salinity at stations I2 and I3 decreased sharply in the upper ~10 cm layer of the sediment. This result could be due to a strong advective flow of fresher groundwater discharged into the zone of stations I2 and I3. Kim (2002) confirmed previously the net flow of coastal groundwater (brackish groundwater) across the sediment-water interface in the same area of this study
using a manual seepage meter. Salinity at stations I1 and I2 located in the intertidal zone showed a large temporal variation over all depths during the half tidal cycle, whereas salinity at stations I3 and I4 located in the sublittoral zone showed only a slight variation with a similar pattern. This implies that the exchange between the pore water and overlying seawater is very active in the intertidal zone (stations I1 and I2) through repeated emersion and immersion, whereas the sublittoral zone (stations I3 and I4) which is continuously submersed is very stable.
The ammonium concentrations in the pore water were vertically uniform with almost negligible concentrations at stations I1 and I2. In general, the repeated emersion and immersion may have an impact on the distribution of dissolved chemical constituents including nutrients in pore water (Dolphin et al. 1995; Kuwae et al. 2003). As such, Rocha (1998) discovered the accumulation of dissolved ammonium in the surface sediment of Sado estuary (western Portugal) during emersion. However, the ammonium concentrations at stations I1 and I2 in this study area did not exhibit the influence of emersion and immersion. This appears to be related to the redox condition in the sediment depending on the exposure time of surface sediment since our pore water samples were collected within 15 minutes after the emersion. According to Rocha (1998), the dissolved and sorbed ammonium pool size increases gradually until the end of exposure, when an abrupt reduction takes place. In addition, Usui et al. (1998) reported that the nitrate concentration of pore water in Tama estuary sediment significantly decreased during the initial 3 to 4 hours after the onset of emersion.
Significant increases of ammonium are shown in the subsurface layer of stations I3 and I4, which showed stable salinity over the daily tidal cycle. In general, the concentration of ammonium in pore water of sediment could increase due to various geochemical processes such as ammonification, advection of pore water, diffusion from sediments, and
biotic processes (i.e. N2-fixation). The increases of ammonium
by diffusion from sediments and biotic processes in the subsurface layer of sediment should be insignificant at stations I3 and I4 based on the vertical profiles of ammonium. Furthermore, if the variation of ammonium concentration is dependent on the advection flow of pore water, the concentration of ammonium in subsurface layer is expected to be almost constant with depth as shown at station I2 in Fig. 3. Thus, these increases appear to be related to the
regeneration of ammonium by the decomposition of organic matter and/or ammonification (nitrate reduction) in the shallow depths of sediments where the water exchange is limited.
Nitrate plus nitrite (NO3+NO2) concentrations in the pore
water at station I1 were relatively uniform with depth, with some temporal variation over all depths. This might be associated with rapid physical ventilation of oxygenated seawater into the entire depth, as indicated by the profiles of temperature and salinity. Although station I2 might also be influenced by such physical ventilations, the nitrate plus nitrite concentrations in the pore water increased gradually with increasing depth. Similarly, increased nitrate concentrations in the subsurface layer (>20 cm depth) are shown at station I3. Thus, the higher nitrate plus nitrite
concentrations in the deeper layer at stations I2 and I3 appear to be due to the influence of fresh groundwater. Most of the nitrogen species occur as ammonium at station I4, with negligible nitrate and nitrite, indicating the occurrence of an anoxic environment in the subsurface layer. On the contrary, the high nitrate plus nitrite concentration in the ~30 cm layer of sediment during low tide appears to be associated with the pore water flow in the subsurface sediment based on the vertical profiles of nutrient concentrations in the pore water. To confirm our hypothesis, more extensive
investigation of chemical signatures (i.e. SF6) in pore water
will be necessary in the future.
The concentrations of DIP and DSi in pore water at stations I2 and I3 were relatively higher than those at stations I1 and I4, with notably higher concentrations at
Fig. 3. Vertical profiles of temperature, salinity, nutrient concentrations in seawater and pore water from the Ilkwang Bay sandflat on August
29 (stations I1 and I3) and September 2 (stations I2 and I4), 2005. Stations I1 and I2 are located in the intertidal zone, whereas stations I3 and I4 are located in the sublittoral zone.
station I3. This appears to be associated with the occurrence of fresher groundwater in the subsurface layer, as shown for nitrate plus nitrite.
Daily variations off the Nakdong River Estuary
Since the salinity of pore water in coastal sediments is generally similar to that of seawater, it is difficult to identify the influence of re-circulating seawater in the sediment column. However, the low-salinity coastal seawater in this study region could be a good indicator of exchange between the pore water and overlying water through water-sediment
interface. At station D2 in the Dadaepo intertidal sandflat, the salinity in the pore water showed a large variation within ~15 cm depth of sediments (Fig. 4). The salinity in the pore water was relatively higher than that of the overlying estuarine seawater except on August 14, indicating an active exchange between the pore water and overlying seawater. The temporal variations of DSi and DIN concentrations in the pore water were significant over the sampling period (Fig. 4). We do not find any distinctive patterns for species either in vertical or temporal trends. The vertical profiles of
nitrate plus nitrite (NO3+NO2) in the pore water decreased
Fig. 4. Vertical profiles of temperature, salinity, and nutrient (silicate, ammonium, and nitrite plus nitrate) concentrations in seawater
with depth, showing a constant concentration below 15 cm depth. Recently, Na and Lee (2005) reported that the decrease of nitrate in the pore water with depth at the intertidal sandflat of Ganghwa Island were due to the consumption of nitrate during the decomposition of organic matter under the anaerobic condition of the subsurface sediment. Therefore, nitrate plus nirite profiles at station D2 may be due to denitrification by the decomposition of organic matter and/or by anaerobic microbial respiration in the subsurface sediments where the water exchange is limited. On the contrary, the vertical profiles of ammonium concentration in the pore water increased with depth, which differs from the profiles in the Ilkwang stations. This appears to be associated with nitrification under the aerobic condition of surface sediment, diffusion flux from surface sediment to overlying estuarine water, and the consumption of benthic phytoplankton in surface sediment after the ammonium is regenerated by the mineralization of organic
nitrogen and/or nitrate reduction (nitrate ammonification) in sediment.
The average salinity in the pore water increased gradually with time but experienced a sharp decline on August 24, 2003 (Fig. 5). Similarly, the average DSi concentration showed a gradual decrease with time throughout the study period. The average DIN concentration in pore water did not show any relation to precipitation and tidal cycle during the study period. The amount of freshwater input through
river discharge was more than 40 × 107
m3 day-1
in the early
study period, decreased suddenly to below 1.0 × 107
m3 day-1
on August 5,and then again increased to above 40 × 107
m3 day-1
on August 18. Salinity and DSi concentrations in coastal seawater and pore water showed a good negative correlation (Fig. 6; r2
= 0.50, p<0.001). This suggests that salinity and DSi concentrations in pore water are related to the amount of river discharge in this study area. Although the salinity in estuarine water was much lower than that in
Fig. 5. Temporal variations of average salinity (upper) and nutrient concentrations (lower) in pore water from the Nakdong River Estuary
sandflat (Dadaepo Beach) from July 29 to August 24, 2003. The scale of the precipitation is not shown on the figure, but the height of the vertical bar is conformable to the same value of the DSi concentration.
pore water of surface sediment during each sampling time, it increased gradually with time during the study periods. In addition, the water loss and temperature rise of surface sediment in the intertidal sandflat during exposure appear to cause the intrusion of lower-salinity estuarine water during a flood tide. Recently, Rocha (1998) suggested that the convective turnover of the pore water in the surface sediment happens when the intertidal sandflat is inundated after exposure and the convection after flooding is supported by water loss and temperature rise of the sediment during exposure and by the sandy characteristics (large porosity) of the sediment. Some researchers also reported that active exchange between seawater and pore water in sandy sediments is enhanced by wave action, tidal effect, and
bio-irrigation etc. (Riedl et al. 1972; Usui et al. 1998; Martin et
al. 2006; Robinson et al. 2007). Thus, based on the overall
distribution patterns of the measured salinity and nutrient concentrations in coastal seawater and pore water, temporal variations of nutrient concentrations in the pore water at the Dadaepo intertidal sandflat seem to be mainly controlled by rapid mixing with overlying estuarine water, most likely due to wave action, tidal fluctuation and density-driven convection (Riedl et al. 1972; Usui et al. 1998; Rocha 2000; Robinson et al. 2007). This seems to be clear evidence of an upper sediment column exchange of water and nutrients due to the intrusion of lower-salinity and high-Si estuarine water.
Spring-neap tide variations on Naro Island
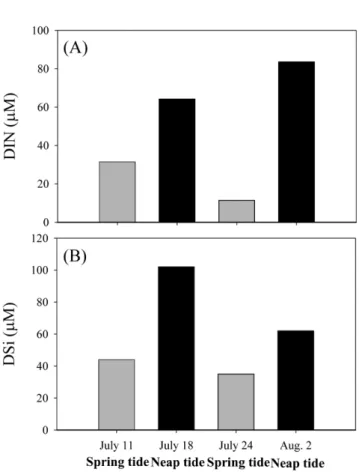
At the Naro Island intertidal sandflat, salinity ranged from 21.5 to 30.9 ppt (mean 27.8 ± 4.3 ppt, n=4), with the lowest value on July 18 compared to other sampling dates (Table 1). The concentrations of DIN ranged from 11 to
84 μM (mean 48 ± 32 μM), and nitrate accounted for the
majority of DIN (> 70%) in sediment pore waters. The
concentrations of DSi ranged from 35 to 102 μM (mean
61 ± 30 μM).
The nutrient concentrations in pore water were higher at the neap tide than at the spring tide (Fig. 7). In general, the pore water of coastal sediments is controlled mainly by tidal fluctuation (Taniguchi 2002; Burnett et al. 2003; Lambert and Burnett 2003). Kim and Hwang (2002) have suggested that SGD increases sharply from neap to spring tide during the wet season, which is regulated predominantly by a semimonthly fluctuation of a tidal oscillating pumping force. In this case, nutrient concentrations in the pore water of coastal sediments can decrease during the spring tide since the exchange of pore water with overlying lower-nutrient seawater is more active. Thus, our results support the previous hypothesis (Kim and Hwang 2002) that the semimonthly variations of SGD driven nutrient fluxes can
Fig. 6. The plots of salinity versus DSi in seawater and pore
water in the Nakdong River Estuary sandflat (Dadaepo Beach) from July 29 to August 24, 2003. Each symbol represents the pore water (single mark) in the surface sediment and seawater (double mark) samples collected for each sampling date.
Table 1. The concentrations of DIN, DIP, and DSi in the pore water around Naro Island during July, 2002
Station Number and Date
Location Temp. (°C) Sal. (ppt) Nutrient concentration (μM) Latitude Longitude NH4 + NO2 − NO3 − DIN DSi St. N1 34°38'50°''N 127°43'50''E
July 11 (Spring Tide) - 28.2 7.5 0.66 23.3 31.5 44
July 18 (Neap Tide) - 21.5 2.6 0.01 61.6 64.2 102
July 24 (Spring Tide) - 30.9 1.5 0.01 9.9 11.4 35
be significant in the coastal ocean.
4. Conclusions
The nutrient concentrations in shallow pore water column of coastal sediments showed dynamic profiles depending on such conditions as fresh groundwater intrusion, overlying seawater, emersion-immersion, and tidal fluctuation. Very complex mixing between porewater and overlying seawater was observed. Hourly, daily, and semi-monthly variations in salinity and nutrient concentrations suggest that the nutrient discharge from coastal sediments is influenced by various oceanographic factors in addition to geological settings. In the future, more extensive studies are necessary to better understand the exchange processes of pore water and seawater using sensitive tracers (i.e. injection of SF6) in the shallow coastal sediments.
Acknowledgements
We thank Y.W. Lee, D.J. Joung, S.I. Ok and D.H. Park who helped with sampling and nutrient analyses. This work was supported by Korea Science and Engineering Foundation (KOSEF, R01-2006-000-10646-0).
References
Boehm, A.B., A. Paytan, G.G. Shellenbarger, and K.A. Davis. 2006. Composition and flux of groundwater from a California beach aquifer: Implications for nutrient supply to the surf zone. Cont. Shelf Res., 26, 269-282.
Burnett, W.C., H. Bokuniewicz, M. Huettel, W.S. Moore, and M. Taniguchi. 2003. Groundwater and pore water inputs to the coastal zone. Biogeochem., 66, 3-33.
Burnett, W.C., P.K. Aggarwal, A. Aureli, H. Bokuniewicz, J.E. Cable, M.A. Charette, E. Kontar, S. Krupa, K.M. Kulkarni, A. Loveless, W.S. Moore, J.A. Oberdorfer, J. Oliveira, N. Ozyurt, P. Povinec, A.M.G. Privitera, R. Rajar, R.T. Ramessur, J. Scholten, T. Stieglitz, M. Taniguchi, and J.V. Turner. 2006. Quantifying submarine groundwater discharge in the coastal zone via multiple methods. Sci. Total Environ., 367, 498-543.
Burnett, W.C., G. Wattayakorn, M. Taniguchi, H. Dulaiova, P. Sojisuporn, S. Rungsupa, and T. Ishitobi. 2007. Groundwater-derived nutrient inputs to the upper Gulf of Thailand. Cont.
Shelf Res., 27, 176-190.
Cable, J.E., J.B. Martin, P.W. Swarzenski, M.K. Lindenberg, and J. Steward. 2004. Advection within shallow pore waters of a coastal lagoon, Florida. Ground Water, 42, 1011-1020. Charette, M.A., K.O. Buesseler, and J.E. Andrews. 2001. Utility
of radium isotopes for evaluating the input and transport of groundwater-derived nitrogen to a Cape Cod estuary. Limnol.
Oceanogr., 46, 456-470.
Charette, M.A. and E.R. Sholkovitz. 2006. Trace element cycling in a subterranean estuary: Part 2. Geochemistry of the pore water. Geochim. Cosmochim. Acta, 70, 811-826.
Charette, M.A., E.R. Sholkovitz, and C.M. Hansel. 2005. Trace element cycling in a subterranean estuary: Part 1. Geochemistry of the permeable sediments. Geochim. Cosmochim. Acta, 69, 2095-2109.
Corbett, D.R., J. Chanton, W.C. Burnett, K. Dillon, C. Rutkowski, and J.W. Fourqurean. 1999. Patterns of groundwater discharge into Florida Bay. Limnol. Oceanogr., 44, 1045-1055. Dolphin, T.J., T.M. Hume, and K.E. Parnell. 1995. Oceanographic
processes and sediment mixing on a sand flat in an enclosed sea, Manukau Harbor, New Zealand. Mar. Geol., 128, 169-181. Garrson, G.H., C.R. Glenn, and G.M. McMurtry. 2003. Measurement of submarine groundwater discharge in Kahana Bay, O‘ahu,
Fig. 7. Temporal variations of (A) DIN and (B) DSi in pore water
from shallow sediments in the Naro Island sandflat, in July 2002. The grey and black bars represent the pore water samples collected during spring and neap tides, respectively. The nutrient concentrations in pore water are relatively higher at neap tide than at spring tide and show a semimonthly variation.
Hawaii. Limnol. Oceanogr., 48, 920-928.
Huettel, M. and I.T. Webster. 2001. Porewater flow in permeable sediments. p.144-179. In: the benthic boundary layer – transport
processes and biogeochemistry, ed. by B.P. Boudreau and
B.B. Jorgensen. Oxford University Press, London.
Hwang, D.W., Y.W. Lee, and G. Kim. 2005. Large submarine groundwater discharge and benthic eutrophication in Bangdu Bay on volcanic Jeju Island, Korea. Limnol. Oceanogr., 50, 1393-1403.
Kelly, R.P. and S. B. Moran. 2002. Seasonal changes in groundwater input to a well-mixed estuary estimated using radium isotopes and implications for coastal nutrient budgets. Limnol.
Oceanogr., 47, 1786-1807.
Kim, G. 2002. Influence of submarine groundwater discharge for marine pollution and red-tide. p. 91-114. In: Proceedings of
the autumn meeting, 2002, the Korean Society of Oceanography,
Seoul.
Kim, G. and D.W. Hwang. 2002. Tidal pumping of groundwater into the coastal ocean revealed from submarine 222
Rn and CH4
monitoring. Geophys. Res. Lett., 29, doi: 10.1029/2002GL015093. Kim, G., J.W. Ryu, H.S. Yang, and S.T. Yun. 2005. Submarine
groundwater discharge (SGD) into the Yellow Sea revealed by 228
Ra and 226
Ra isotopes: Implications for global silicate fluxes. Earth Planet. Sci. Lett., 237, 156-166.
Krest, J.M., W.S. Moore, L.R. Gardner, and J.T. Morris. 2000. Marsh nutrient export supplied by groundwater discharge: Evidence from radium measurements. Global Biogeochem.
Cycles, 14, 167-176.
Kuwae, T., E. Kibe, and Y. Nakamura. 2003. Effect of emersion and immersion on the porewater nutrient dynamics of an intertidal sandflat in Tokyo Bay. Estuar. Coast. Shelf Sci., 57, 929-940.
Lambert, M. and W.C. Burnett. 2003. Submarine groundwater discharge estimates at a Florida coastal site based on continuous radon measurements. Biogeochem., 66, 55-73.
Martin, J.B., J.E. Cable, J. Jaeger, K. Hartl, and C.G. Smith. 2006. Thermal and chemical evidence for rapid water exchange across the sediment-water interface by bioirrigation in the Indian River Lagoon, Florida. Limnol. Oceanogr., 51, 1332-1341.
Martin, J.B., J.E. Cable, P.W. Swarzenski, and M.K. Lindenberg.
2004. Enhanced submarine ground water discharge from mixing of pore water and estuarine water. Ground Water, 42, 1000-1010.
Na, T.H. and T. Lee. 2005. Estimation of denitrification in the Ganghwa tidal flat by a pore water model. J. Kor. Soc.
Oceanogr. (The Sea), 10, 56-68.
Niencheski, L.F.H., H.L. Windom, W.S. Moore, and R.A. Jahnke. 2007. Submarine groundwater discharge of nutrients to the ocean along a coastal lagoon barrier, Southern Brazil.
Mar. Chem., 106, 546-561.
Precht, E. and M. Huettel. 2003. Advective pore-water exchange driven by surface gravity waves and its ecological implications.
Limnol. Oceanogr., 48, 1674-1684.
Riedl, R., N. Huang, and R. Machan. 1972. The subtidal pump: a mechanism of intertidal water exchange by wave action.
Mar. Biol., 13, 210-221.
Robinson, C., B. Gibbes, H. Carey, and L. Li. 2006. Driving mechanisms for flow and salt transport in a subterranean estuary. Geophys. Res. Lett., 33, doi: 10.1029/2005GL025247. Robinson, C., L. Li, and D.A. Barry. 2007. Effect of tidal forcing on a subterranean estuary. Advan. Water Res., 30, 851-865. Rocha, C. 1998. Rhythmic ammonium regeneration and flushing
in intertidal sediments of the Sado estuary. Limnol. Oceanogr.,
43, 823-831.
Rocha, C. 2000. Density-driven convection during flooding of warm, permeable intertidal sediments: the ecological importance of the convective turnover pump. J. Sea Res., 43, 1-14. Shum, K.T. and B. Sundby. 1996. Organic matter processing in
continental shelf sediments-the subtidal pump revisited.
Mar. Chem., 53, 81-87.
Swarzenski, P.W., C. Reich, K.D. Kroeger, and M. Baskaran. 2007. Ra and Rn isotopes as natural tracers of submarine groundwater discharge in Tampa Bay, Florida. Mar. Chem.,
104, 69-84.
Taniguchi, M. 2002. Tidal effects on submarine groundwater discharge into the ocean. Geophys. Res. Lett., 29, doi: 10.1029/2002GL014987.
Usui, T., I. Koike, and N. Ogura. 1998. Tidal effect on dynamics of pore water nitrate in intertidal sediment of a eutrophic estuary. J. Oceanogr., 54, 205-216.