이
이
이학
학
학 석
석
석사
사
사학
학
학위
위
위 논
논
논문
문
문
N
N
Ne
e
eu
u
ur
r
ro
o
op
p
pr
r
ro
ot
o
t
te
e
ec
c
ct
ti
t
i
io
o
on
n
no
o
of
f
fA
A
AG
G
G4
4
49
9
90
0
0i
i
in
n
nO
Ox
O
x
xi
i
id
d
da
at
a
t
ti
i
iv
v
ve
e
e
S
S
St
t
tr
r
re
e
es
s
ss
s
s-
-
-i
i
in
n
nd
d
du
u
uc
c
ce
e
ed
d
dN
N
Ne
e
eu
u
ur
r
ro
o
on
n
na
a
al
l
lD
D
De
e
ea
a
at
t
th
h
h
아
아
아 주
주
주 대
대
대 학
학
학 교
교
교 대
대
대 학
학
학 원
원
원
의
의
의 학
학
학 과
과
과
김
김
김 아
아
아 영
영
영
N
N
Ne
e
eu
u
ur
r
ro
o
op
p
pr
r
ro
o
ot
t
te
e
ec
c
ct
t
ti
i
io
o
on
n
no
o
of
f
fA
A
AG
G
G4
4
49
9
90
0
0i
i
in
n
nO
Ox
O
x
xi
i
id
d
da
at
a
t
ti
i
iv
v
ve
e
e
S
S
St
t
tr
r
re
e
es
s
ss
s
s-
-
-i
i
in
n
nd
d
du
u
uc
c
ce
e
ed
d
dN
N
Ne
e
eu
u
ur
r
ro
o
on
n
na
a
al
l
lD
D
De
e
ea
a
at
t
th
h
h
b
b
by
y
y
A
A
Ah
h
hY
Y
Yo
o
ou
u
un
n
ng
g
gK
K
Ki
i
im
m
m
A
A
A D
D
Di
i
is
s
ss
s
se
er
e
r
rt
t
ta
a
at
t
ti
i
io
on
o
n
nS
S
Su
u
ub
b
bm
m
mi
i
it
t
tt
t
te
e
ed
d
dt
t
to
oT
o
T
Th
h
he
e
eG
G
Gr
r
ra
a
ad
d
du
u
ua
a
at
t
te
e
eS
S
Sc
c
ch
h
ho
o
oo
o
ol
l
lo
o
of
f
fA
A
Aj
j
jo
o
ou
u
u
U
U
Un
n
ni
i
iv
v
ve
e
er
r
rs
s
si
i
it
t
ty
y
yi
i
in
n
nP
P
Pa
a
ar
r
rt
t
ti
i
ia
a
al
l
lF
F
Fu
ul
u
l
l
f
f
fi
i
il
l
ll
l
lm
m
me
e
en
n
nt
t
to
o
of
f
ft
t
th
he
h
e
eR
R
Re
e
eq
q
qu
u
ui
i
ir
r
re
e
em
m
me
e
en
n
nt
t
ts
s
sf
f
fo
o
or
r
rt
t
th
h
he
e
e
D
D
De
e
eg
g
gr
r
re
e
ee
e
eo
o
of
f
f
M
M
MA
A
AS
S
ST
T
TE
E
ER
R
R O
O
OF
F S
F
S
SC
C
CI
I
IE
E
EN
N
NC
C
CE
E
E
S
S
Su
u
up
p
pe
e
er
r
rv
v
vi
i
is
s
se
e
ed
d
db
b
by
y
y
E
E
Eu
u
un
n
nJ
J
Jo
o
oo
o
oB
B
Ba
a
ai
i
ik
k
k,
,
,M
M
M.
.
.
D
D
D.
.
.
,
,
,P
P
Ph
h
h.
.
.
D
D
D.
.
.
D
D
De
e
ep
p
pa
a
ar
r
rt
t
tm
m
me
e
en
n
nt
t
to
o
of
f
fM
M
Me
e
ed
d
di
i
ic
c
ca
a
al
l
lS
S
Sc
ci
c
i
ie
e
en
n
nc
c
ce
e
es
s
s
T
T
Th
h
he
e
eG
G
Gr
r
ra
a
ad
d
du
u
ua
a
at
t
te
e
eS
S
Sc
c
ch
h
ho
o
oo
o
ol
l
l,
,
,A
A
Aj
j
jo
o
ou
u
uU
U
Un
n
ni
i
iv
v
ve
e
er
r
rs
s
si
i
it
t
ty
y
y
F
F
Fe
e
eb
b
br
r
ru
u
ua
a
ar
r
ry
y
y,
,
,2
2
20
0
00
0
07
7
7
김
김
김아
아
아영
영
영의
의 이
의
이
이학
학
학 석
석
석사
사
사학
학
학위
위
위 논
논
논문
문
문을
을 인
을
인
인준
준
준함
함
함.
.
.
심
심
심사
사
사위
위
위원
원
원장
장
장
문
문
문 창
창
창 현
현
현
인
인
인
심
심
심 사
사
사 위
위
위 원
원
원
이
이
이 수
수
수 환
환
환
인
인
인
심
심
심 사
사
사 위
위
위 원
원
원
백
백
백 은
은
은 주
주
주
인
인
인
아
아
아 주
주
주 대
대
대 학
학
학 교
교
교 대
대
대 학
학
학 원
원
원
2
2
20
0
00
0
06
6
6년
년
년 1
1
12
2
2월
월
월 2
2
22
2
2일
일
일
감 감감사사사의의의 글글글 확고했던 다짐이 망설임으로 바뀌면서 2년이란 시간이 지나 미완성작 같 은 논문을 쓰게 되었습니다.독립하지도 못한 딸에게 2년 동안 가장 많은 치다꺼 리를 해주신 부모님께 감사의 말씀 올립니다.첫 면접에서 씩씩한 모습이 좋았다 며,함께 공부할 수 있는 기회를 주신 백은주 교수님 감사합니다.여전히 씩씩하 긴 한데,여전히 마음에 드시는지 모르겠습니다.새로운 실험을 할 때마다 실험 의 기본과 목적에 대해서 잊지 않도록 도와주신 이수환 교수님,선생님의 관심과 애정에 감사드립니다.과학자로서의 자세만큼이나 실험실 가족들과의 단합을 중 요하게 여기시는 문창현 교수님,재미있는 생리학교실이라서 실험하는 것이 즐겁 습니다.감사합니다.연구하는 사람의 열정을 제대로 보여주신 정이숙 교수님,지 금 함께하지 못하는 것이 아쉽습니다.제가 하고 있는 일을 선행해 오신 권경자 선생님,선생님의 부재로 혼자서 가는 길이 캄캄합니다.그립습니다.이제 곧 미 국에서 새로운 둥지를 틀 박지영 선생님,천방지축 후배를 귀엽게 봐주셔서 감사 합니다.보고 싶을 겁니다.생리학교실에 처음 온 날부터 맺어온 청소 짝지의 인 연을 이어가고 있는 장유나 선생님,실험실 생활을 성실히 할 수 있는 것은 선생 님의 덕분입니다.고맙습니다.건강한 아기의 엄마가 되어 돌아오실 김윤희 선생 님,선생님만큼 자기 일에 욕심 있는 사람이 되고 싶습니다.동기라는 이유만으 로도 든든한 힘이 되어준 김희재,덕분에 실험실에 빨리 적응할 수 있었어.옆에 서 시끄럽게 해서 미안해.한 학기 늦게 들어온 이유로 아직도 궂은일을 하고 있 는 정지인,다음 학기에는 선생님이 졸업준비로 바쁠 때 내가 꼭 도와줄께요.같 이 고생하며 졸업하게 된 윤이슬,졸업을 진심으로 축하한다.취업하면 주전부리 사들고 학교에 자주 놀러오기로 한 것 잊으면 안돼.마음고생이 많은 안진희, 옆에서 투정부리는 것 받아줘서 고마워.내년에는 내가 다 받아줄게.모든 실험 실 선생님,석사 생활을 하면서 많은 도움을 받았습니다.고맙습니다.마지막으로 실험실의 가장 중앙에 앉아 있는 박은남,실험실의 모든 살림을 잘 꾸려줘서 진 심으로 고마워.
- ABSTRACT -
Neuroprotection of AG490 in Oxidative Stress-induced Neuronal
Death
Oxidative stress induced increases of the free radicals, which has an important role in neuronal death. Free radicals oxidize intracellular molecules, the destabilization of membrane integrity, and the mitochondrial dysfunction. In present study, we investigated whether AG490, a specific Janus kinases 2 (JAK2) inhibitor had an effect on oxidative stress-induced cytotoxicity in cultured primary cortical neurons.
AG490 significantly protected neuronal cell death against various oxidative stimuli, such as hydrogen peroxide (H2O2), L-buthionin-S,R-sulfoxamine (BSO),
N-methyl-D-aspartic acid (NMDA), and arachidonic acid (AA). Particularly, AG490 completely blocked H2O2-induced neuronal death. To explore the protective mechanism of AG490, the
involvement of JAK2 signal pathway, the regulation of reactive oxygen species (ROS), and the effect on mitochondrial functions were examined. AG490 suppressed oxidant-induced JAK2 phosphorylation. In addition, AG490 regulated the increase of intracellular ROS by oxidative stress and AG490 itself has a modest antioxidant action and increased the total glutathione (GSH) level. Furthermore, AG490 significantly abolished the reduction of mitochondrial membrane potential by oxidative stress.
Taken together, AG490 had a potent protective effect in oxidative stress-induced neuronal death. The protection mechanism of AG490 can be explained with involvement of JAK2 pathway, directly scavenging ROS and up-regulation of endogenous antioxidants activity, and restored mitochondrial functions.
TABLE OF CONTENTS
ABSTRACT--- ⅰ TABLE OF CONTENTS---ⅲ LIST OF FIGURES--- ⅴ LIST OF TABLES--- ⅵ ABBREVIATION---ⅶ Ⅰ. INTRODUCTION---1Ⅱ. MATERIALS AND METHODS--- 4
A. MATERIALS---4
B. METHODS--- 5
1. CELL CULTURE--- 5
2. ASSESSMENT OF NEURONAL DEATH--- 6
3. DPPH RADICAL SCAVENGING ACTIVITY--- 6
4. ABTS RADICAL CATION DECOLORIZATION ASSAY---7
5. FERROUS METAL CHELATING ACTIVITY--- 7
6. REDUCING POWER---8
7. MEMBRANE LIPID PEROXIDATION---8
8. WESTERN BLOT ANALYSIS TO CONFIRM JAK/STAT PATHWAY---9
9. MEASUREMENT OF ROS FORMATION---9
10. MEASUREMENT OF MITOCHONDRIAL MEMBRANE POTENTIAL---10
11. GSH AND GSSG ASSAYS---10
12. STATISTICAL ANALYSIS---12
Ⅲ. RESULTS---13
A. AG490 ATTENUATES NEURONAL CELL DEATH AGAINST OXIDATIVE STRESS---13
B. ANTIOXIDANTS HAVE DIFFERENTIAL EFFECTS ON OXIDATIVE NEURONAL CELL DEATH---15
C. AG490 DECREASES THE PHOSPHORYLATION OF JAK2 AGAINST OXIDATIVE STRESS--- 17
D. OXIDATIVE STRESS-INDUCED INTRACELLULAR ROS PRODUCTION IS REGULATED BY AG490 AND ANTIOXIDANTS--- 20
E. ANTIOXIDANT ACTIVITIES IN VITRO---25
F. AG490 INCREASES THE INTRACELLULAR GSH LEVEL IN CORTICAL NEURONS--- 32
G. OXIDATIVE STRESS DISTURBED THE MITOCHONDRIAL MEMBRNE POTENTIAL IN CORTICAL NEURONS---34
Ⅳ. DISCUSSION---39
Ⅴ. CONCLUSION---44
REFERENCES---45
LIST OF FIGURES
Fig. 1. Protective effects of AG490 in oxidative stress-induced neuronal death---14
Fig. 2. Effects of antioxidants in oxidative stress-induced neuronal death---16
Fig. 3. Inhibition of oxidative stress-induced JAK2 phosphorylation by AG490---18
Fig. 4. ROS generation against hydrogen peroxide (H2O2) in cortical neurons---21
Fig. 5. ROS generation against buthionin sulfoxamine (BSO) in cortical neurons---23
Fig. 6. DPPH radical scavenging activity---27
Fig. 7. ABTS radical cation decolorization assay---28
Fig. 8. Metal chelating activity--- 29
Fig. 9. Reducing power--- 30
Fig. 10. Lipid peroxidation with C11-BODIPY581/591--- 31
Fig. 11. Reduction of mitochondrial membrane potential (MMP) against H2O2 in cortical neurons---35
Fig. 12. Reduction of mitochondrial membrane potential (MMP) against BSO in cortical neurons---37
LIST OF TABLES
ABBREVIATION
AG490: Tyrphostin B42, α-Cyano-(3, 4-dihydroxy)-N-benzylcinnamide JAK: Janus kinase
H2O2: Hydrogen peroxide
BSO: Buthionin sulfoxamine NMDA: N-methyl-D-aspartic acid AA: Arachidonic acid
LDH: Lactate dehydrogenase
Trolox: 6-Hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid NAC: N-acetyl-L-cysteine
BHT: Butylated hydroxytoluene GSH: reduced glutathione GSSG: oxidized glutathione ROS: Reactive oxygen species
MMP: Mitochondrial membrane potential DPPH: 2,2-Diphenyl-1-picrylhydrazyl
ABTS: 2-azino-bis(3-ethylbenzenthiazoline-6-sulfonic acid) H2DCF-DA: 2’,7’-dichlorofluorescein diacetate
. I
Ⅰ
Ⅰ
Ⅰ
Ⅰ NTRODUCTION
Brain activity is mainly maintained with the consumption of energy produced through aerobic metabolism with oxygen. However, the metabolism also generates by-products, such as reactive oxygen species (ROS) and reactive nitrogen species (RNS). When reactive species are excessively produced, these oxidize the intracellular lipids, proteins and nucleic acids, and result in oxidative damage; caused by direct attack of reactive species during oxidative stress. Besides oxidative damage, oxidative stress leads to changes of ion levels or activation of proteases, finally triggers cell death and develops various neurodegenerative diseases including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease and amyotrophic lateral sclerosis.
Oxidative stress induces lipid peroxidation and thereby causes malfunction of membranes by altering stability of the cell membrane and activity of the membrane-bound enzymes and receptors. Membrane stabilization by vitamin E analogs results in an up-regulation of intracellular signaling and finally potentiates the capacity to resist oxidative stress. Normally, brain preserves a high ratio of GSH/GSSG for antioxidant defense. Loss of total glutathione and a low ratio of GSH/GSSG are markers of oxidative stress. Main neuronal defense against hydrogen peroxide generated in the mitochondria is mediated by glutathione system. Decrease in the cellular GSH results in accumulation of ROS generated by mitochondria as long as oxidized GSH does not rapidly convert into reduced GSH by glutathione peroxidase (GPx). The accumulation of ROS then causes Ca2+ influx from extra-cellular medium. Ca2+ overload induces the opening mitochondrial permeability transition
(MPT) pore that allows flux of solutes from the matrix to inter-membrane space. Subsequently, mitochondria get swelling and appear disruption of the outer mitochondrial membrane leading to release of pro-apoptotic proteins, loss of the proton gradient, and failure of oxidative phosphorylation resulting in decreased ATP production and eventually cellular death.
Tyrphostins are a family of structurally phenolic compounds that have an inhibitory activity of protein tyrosine kinases (PTKs). Therefore tyrphostins affect not only PTKs but also PTKs-independent biological functions because the structures closely resemble those of antioxidants such as vitamin E and propyl gallate. Two main structural determinants in tyrphostins contribute to PTKs-independent functions; hydroxyl group and hydrophobicity. The hydroxyl group on the benzene ring of tyrphostin gives antioxidant activity. Hydrophobicity of tyrphostins enhances an ability to easily penetrate lipid bilayer in order to reach the sites of ROS generation and accumulation.
In this study, the mechanism of AG490-mediated protective consequence against oxidative stress in cultured primary cortical neurons was explained. To explore neuroprotective effects of AG490, the effects on the situation of ROS accumulation and mitochondrial dysfunction induced by oxidative stress were examined. We also compared the effect of exogenous antioxidants, such as trolox and NAC in this model. AG490 decreased the intracellular ROS level generated by oxidative stress, in addition AG490 preserved the mitochondrial membrane potential. However, certain exogenous antioxidant did not block effectively the intracellular ROS production and the abolishment of mitochondrial membrane potential generated by oxidative stress, and then the neuronal death appeared.
.
Ⅱ
Ⅱ
Ⅱ
Ⅱ MATERIALS AND METHODS
A. Materials
Pregnant ICR mouse were obtained from Folas (Seoul, Korea). Minimum essential medium (MEM) was purchased from WELGENE Bioscience (Seoul, Korea), fetal bovine sesum (FBS), horse serum (HS), and L-glutamine were obtained from Gibco-BRL (Gaithersburg, MD). Cytosine-β-arabinofuranoside (Ara-C), L-glucose, hydrogen peroxide (H2O2), buthionin sulfoxamine (BSO), N-methyl-D-aspartic acid (NMDA), arachidonic acid
(AA), triton X-100, phenylmethylsulfonyl fluoride (PMSF), sodium fluoride (NaF), sodium orthovanadate (Na3VO4), EGTA, EDTA, triethanolamine (TEA), 5,
5’-dithiobis(2-nitrobenzoic acid), β-Nicotinamide adenine dinucleotide 2’-phosphate (β-NADPH), oxidized glutathione (GSSG), reduced glutathione (GSH), glutathione reductase (GR), vinylpyridine, 2,2-diphenyl-1-picrylhydrazyl (DPPH), and 2-azino-bis(3-ethylbenzenthiazoline-6-sulfonic acid) (ABTS) were purchased from Sigma (St. Louis, MO, USA). Tyrphostin B42 (AG490) was from Calbiochem (La Jolla, CA). 2’, 7’-dichlorodihydrofluorescein diacetate (H2
DCF-DA), rhodamine 123 and pluronic F-127 were from Molecular Probes (Eugene, OR). Antibodies against phospho-JAK2 and JAK2 were from Upstate (Milton Keynes, UK). Anti-rabbit horseradish peroxidase was purchased from Cell Signaling Technology (Beverly, MA, USA).
B. Methods
1. Cell Culture
Neuronal cultures were obtained from dissociated embryonic day (E14) cortices of ICR mouse brains. Briefly, cortices freed of meninges were mechanically dissociated and gently triturated twice with a flame-polished Pasteur pipette. Cells were plated in poly-D-lysine precoated 6-well and 24-well plates and maintained in culture medium (minimal essential medium supplemented with 20mM glucose) at 37℃ in a humidified atmosphere of 5% CO2/95% air. To inhibit the growth of glial cells, 5µM Ara-C was added within 48h - 72h
as previously described (Choi et al., 1987). The culture medium was supplemented with 5% fetal bovine serum, 5% horse serum and 2mM L-glutamine during 7 days, which time neurons needed to differentiate from neuronal precursor cells. Differentiated neurons were identified with a monoclonal antibody against the neuron-specific nuclear protein, NeuN (Chemicon, Temecula, CA, USA). For immunostaining, cells were fixed with 4% paraformaldehyde in phosphate buffered saline (PBS; 0.01M pH 7.4). After washing with PBS, cells were blocked in 0.5% BSA in PBS, and then incubated with the primary antibody against NeuN (1:100, Chemicon, Temecula, CA, USA) in PBS containing 0.1% BSA for 2h at 37oC. After several washes in PBS, the cells were incubated with biotinylated anti-rabbit and anti-mouse secondary antibodies for 1h at 37oC and staining was visualized by the biotin-streptavidin peroxidase method (LSAB kit, DAKO, Carpinteria, CA, USA). All cultures used in the experiments were 7d old.
2. Assessment of neuronal death
H2O2 (50µM), buthionin sulfoxamine (BSO, 1mM), NMDA (100 µM), and AA (50
µM), were used to induce the neuronal toxicity. In all experiments, cells were treated with JAK2 inhibitor 30 minutes prior to administration of various stimuli.
Cellular cytotoxicity was also assessed by measuring the relative amount of lactate dehydrogenase (LDH) released into the culture medium 12-24 hr/ 24-36hr after stimuli. LDH activity in the culture media was measured by absorbance at 340 nm during the conversion of enzyme cofactor NADH to NAD+. Briefly, 25 µl aliquots of culture media were collected and dissolved in NADH solution (0.25 mg in PBS). Pyruvate buffer (22.7 mM sodium pyruvate in PBS, 0.1 M, pH = 7.4) was then added to the samples. Maximal neuronal LDH release was measured following exposure to 500µM NMDA for 6hr. Values were expressed relative to the measurements from maximal neuronal LDH release.
3. DPPH radical scavenging activity
The free radical scavenging activity was measured by 2,2-diphenyl-1-picryl-hydrazil (DPPH) using the method of Shimada et al. (1992). Briefly, 0.1mM solution of DPPH in ethanol was prepared. Then, 0.2ml of this solution was added to 0.6ml of antioxidant at different doses. The mixture was shaken vigorously and allowed to stand at room temperature for 30 min. Then the absorbance was measured at 517 nm in a spectrophotometer. Lower absorbance of the reaction mixture indicated higher free radical
scavenging activity.
4. ABTS radical cation decolorization assay
The spectrophotometric analysis of ABTS radical scavenging activity was determined according to the method of Re et al. (1999). The ABTS cation radical was produced by the reaction between 7mM ABTS in H2O and 2.45mM potassium persulfate,
stored in the dark at room temperature for 12h. Before usage, the ABTS solution was diluted to get an absorbance of 0.700 ± 0.025 at 734 nm with phosphate buffer (0.1M, pH 7.4). Then, 1ml of ABTS solution was added 3ml of antioxidant solution in ethanol at different concentration. After 30 min, the percentage inhibition at 734nm was calculated for each concentration relative to a blank absorbance (ethanol).
5. Ferrous metal chelating activity
The chelating of ferrous ions by reference antioxidants and standard molecules was estimated by the method of Dinis et al. (1994). Briefly, antioxidant in o.4ml was added to a solution of 2mM FeCl2 (0.05ml). The reaction was initiated by the addition of 5mM
ferrozine (0.2ml) and total volume was adjusted to 4ml with ethanol. Then, the mixture was shaken vigorously and left at room temperature for 10 min. Absorbance of the solution was measured spectrophotometrically at 562 nm.
6. Reducing power
The reducing power of antioxidant was determined according to the method of Oyaizu et al. (1986). The different doses of antioxidant in 1ml of distilled water were mixed with phosphate buffer (2.5ml, 0.2M, pH 6.6) and potassium ferricyanide [K3Fe(CN)6] (2.5ml,
1%). The mixture was incubated at 50 ℃ for 20min. A portion (2.5ml) of TCA (10%) was added to the mixture, which was then centrifuged for 10 min at 1000 * g. The upper layer of solution (2.5ml) was mixed with distilled water (2.5ml) and FeCl3 (0.5ml, 0.1%), and the
absorbance was measured at 700 nm in a spectrophotometer. Higher absorbance of the reaction mixture indicated greater reducing power.
7. Membrane lipid peroxidation
Lipid peroxidation was determined using the fluorescent fatty acid analog C11-BODIPY581/591 prepared in DMSO (2mM) and stored under nitrogen at -20 ℃. BODIPY was incorporated into the cell membranes (4 µM in PBS) and incubated for 1h at 37 . The ℃
cells were then washed twice with PBS, and incubated with PBS (control sample), with the oxidant H2O2 or with H2O2 in the presence of antioxidants. At the end of the incubation
period (6h), the membrane lipid oxidation was determined by calculating the ratio between the red fluorescence (580/610 nm) decay of BODIPY and the green fluorescence (485/535 nm) increase of the oxidation product, normalized in respect to the protein content.
8. Western blot analysis to confirm JAK/STAT pathway
Phosphorylation of JAK2 and STAT1/3 in cultured cortical neurons was examined by western blotting (Yadav A, et al., 2005). The media were aspirated, and the cells were washed with ice-cold PBS. Subsequently, the cell was lysed in ice-cold Triton lysis buffer (50mM Tris-HCl, pH7.5, 1% triton, 2mM Na3VO4,, 2mM EGTA, 10mM EDTA, 100mM
NaF, 1mM Na4P2O7 , 100µg/ml phenylmethylsulfonyl fluoride, and 1µg/ml aprotinin,
pepstatin A, and leupeptin). The supernatants collected after removing insoluble debris by centrifuging the samples at 12,000*g for 10min were used in all studies. Proteins concentrations were determined by Pierce BCA assay kit. 100µg of proteins samples were separated by 8% SDS-PAGE and transferred to polyvinylidene difluoride (PVDF, Millipore) membrane. The membrane was incubated with primary antibodies (phospho-JAK2: 1:1000, phospho-STAT1/3: 1:1000), followed by incubation with horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin G secondary antibody at a dilution of 1:5000. The blots were detected using a chemiluminescence western blot kit (ECL; Intron, Korea).
9. Measurement of ROS formation
Intracellular ROS formation was measured fluorometrically using 2’,7’-dichlorofluorescein diacetate (H2DCF-DA) (Lebel et al., 1990). The non-fluorescent dye
DCF-DA permeated cells easily and hydrolyzed to fluorescent 2’, 7’-dichlorofluorescein (DCF) upon interaction with intracellular ROS. In brief, cultured cells were washed with
Hank’s balanced aqueous salts solution (HBSS) buffer containing 120 mM NaCl, 5 mM KCl, 1.6 mM MgCl2, 2.3 mM CaCl2, 15 mM glucose, 20 mM HEPES, and 10 mM NaOH, loaded
with 10µM of DCF-DA and 20% Pluronic F-127 for 30 min, washed again with HBSS. The DCF fluorescence was analyzed using a Spectra MAX Gemini EM (Molecular devices) at an excitation of 490 nm and an emission of 530 nm and a fluorescence microscope.
10. Measurement of mitochondrial membrane potential (∆Ψm)
∆Ψm was assessed by measuring the accumulation of rhodamine 123 (Molecular
probes, Eugene, OR, USA), a membrane-permeable cationic fluorescent dye (Emaus et al., 1986). Cultured cells were loaded with 2uM rhodamine 123 in HEPES control salt solution (HCSS). The cells were incubated for 30 min at 37 ℃ and washed twice with HCSS. The cells were observed with a fluorescence microscope, the fluorescence intensity of rhodamine 123 was quantified using image-analysis computer software.
11. GSH and GSSG assays
GSH and GSSG were determined enzymatically by using a modification of the procedure of Tietze (Tietze et al., 1969) with modifications (Ishige et al., 2001). The method is based on the determination of a chromophoric product, 2-nitro-5-thiobenzoic acid, resulting from the reaction of 5, 5’-dithiobis-(2-nitrobenzoic acid) with GSH. In this reaction, GSH is oxidized to GSSG, which is then reconverted to GSH in the presence of GR and
NADPH. The rate of 2-nitro-5-thiobenzoic acid formation, which is proportional to the sum of GSH and GSSG present, is followed at 405nm.
Measurement of total glutathione
Cells were washed twice with ice-cold phosphate-buffered saline, collected by scraping, and lysed with 3% sulfosalicylic acid. Lysates were incubated on ice for 10min, and supernatants were collected after centrifugation. Upon neutralization of the supernatant with triethanolamine, the concentration of total glutathione (oxidized and reduced) was determined. Briefly, a neutralized supernatant (25µl)was mixed with 175µl of a reaction mixture containing 143mM sodium phosphate (pH 7.5), 6.3mM Na4EDTA, 6mM 5,
5’-dithiobis(2-nitrobenzoic acid), and 0.25mg/ml NADPH. The reaction was started with adding 1 unit/ml glutathione reductase. Color development was monitored at 405nm in a kinetic mode with a microplate reader. Pure GSH was used to obtain a standard curve. The protein content of each sample was determined using the BCA protein assay kit from Pierce with bovine serum albumin as a standard.
Measurement of GSSG
Sample was prepared rapidly to minimize GSH oxidation. Freshly obtained supernatants were treated with 2-vinylpirydine for destroying reduced form of glutathione (Floreani et al., 1997). Typically, supernatant was added 2-vinylpirydine at room temperature for 60 min. TEA was then added; the mixture was vigorously mixed and the pH checked (generally between 6 and 7). Content of GSSG was determined as described above. GSSG
was quantified from a standard curve obtained by plotting known amounts of GSSG.
Measurement of GSH
GSH present in the sample was calculated as the difference between total GSH and GSSG levels, taking into account the fact that one molecule of GSSG gives rise to two molecules of GSH upon reaction with GR.
12. Statistical analysis
Data were expressed as the mean ± standard error of the mean (S.E.M.). All of the experiments were performed from three to six independent experiments. Statistical analysis among groups was performed using one-way analysis of variance (ANOVA) and Student’s t- test. In all cases, values of p are as follows: *p < 0.05, **p < 0.01.
.
Ⅲ
Ⅲ
Ⅲ
Ⅲ RESULTS
A. AG490 attenuated neuronal cell death against oxidative stress
The susceptibility of primary cortical neurons to oxidative stress was evaluated by performing lactate dehydrogenase (LDH) release assay following various stimuli: H2O2
(hydrogen peroxide, 50 µM), BSO (L-buthonine-S,R-sulfoxamine, an inhibitor of γ-glutamylcysteine ligase, 1mM), NMDA (N-methyl-D-aspartate, 100 µM), and AA (arachidonic acid, 50 µM) (Fig. 1). There was the significant neuronal death by all stimuli at 24h. Tyrphostin B42 (AG490), a JAK2 inhibitor (10 µM) was pre-treated 30 min before stimuli administration and prevented cortical neurons from oxidative neuronal death. When AG490 (3-30µM) was incubated against H2O2 and BSO, AG490 also prevented cortical
0 20 40 60 80 100 120 H2O2 BSO NMDA AA L D H r el ea se ( 2 4 h ) (% o f m a x im a l d ea th ) CTL injury AG490 * * * * # # # # (A) 0 20 40 60 80 100 120 H2O2 BSO NMDA AA L D H r el ea se ( 2 4 h ) (% o f m a x im a l d ea th ) CTL injury AG490 * * * * # # # # (A) (B) 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 3uM AG490+H 10uM AG490+H 30uM AG490+H * * # # # # # # 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 3uM AG490+B 10uM AG490+B 30uM AG490+B * * # # # (B) 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 3uM AG490+H 10uM AG490+H 30uM AG490+H * * # # # # # # (B) 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 3uM AG490+H 10uM AG490+H 30uM AG490+H * * # # # # # # 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 3uM AG490+B 10uM AG490+B 30uM AG490+B * * # # # 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 3uM AG490+B 10uM AG490+B 30uM AG490+B CTL 1mM BSO 3uM AG490+B 10uM AG490+B 30uM AG490+B * * # # #
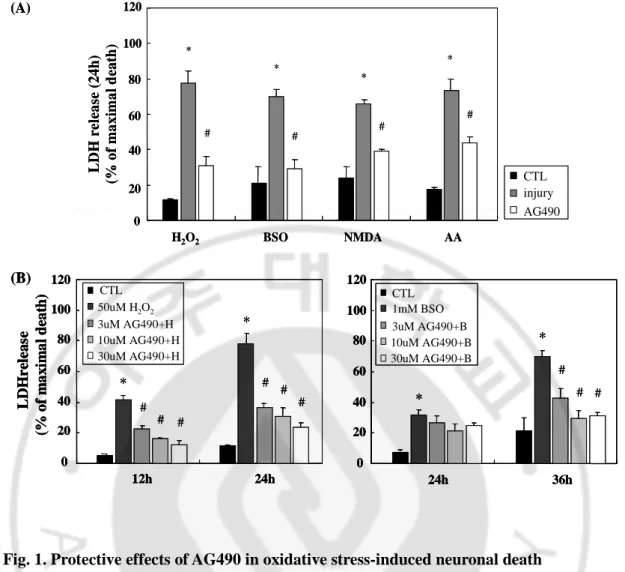
Fig. 1. Protective effects of AG490 in oxidative stress-induced neuronal death
(A) Primary cortical neurons were incubated with AG490 (10μM) 30 min before various stimuli, such as H2O2 (50μM), BSO (1mM), NMDA (100μM), and arachidonic acid (AA,
50μM). Neuronal death was estimated by detecting released lactate dehydrogenage (LDH) at 340nm. (B) AG490 (3-30μM) was pre-treated against H2O2 and BSO. Data are
representatives of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
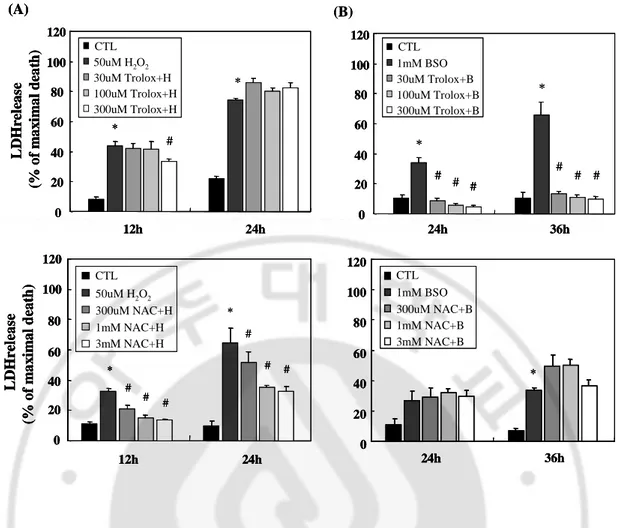
B. Antioxidants had differential effects on oxidative neuronal cell death
To identify neuroprotective mechanisms of AG490 against reactive oxygen species (ROS)-generated cytotoxicity, we compared the effects of broadly used antioxidants, trolox and NAC. Trolox, an α-tocopherol (vitamin E) derivative, is one of the most powerful antioxidants with relatively high selectivity for scavenging peroxynitrite and hydroxyl radical. NAC (N-acetyl-L-cysteine) is a potent antioxidant that is known to increase the intracellular store of glutathione. The neuronal cells were incubated with each antioxidant for 30min prior to 50µM H2O2 or 1mM BSO treatment. Trolox (30µM-300µM) did not prevent
H2O2-induced neuronal cell death, but significantly decreased BSO-induced neuronal cell
death in a concentration-dependent manner. NAC (300µM-3mM) attenuated dose-dependently neuronal death against H2O2, but did not do against BSO (Fig. 2).
0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 30uM Trolox+H 100uM Trolox+H 300uM Trolox+H * * # (A) 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 30uM Trolox+B 100uM Trolox+B 300uM Trolox+B * * # # # # # # (B) 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 30uM Trolox+H 100uM Trolox+H 300uM Trolox+H * * # (A) 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 30uM Trolox+H 100uM Trolox+H 300uM Trolox+H * * # (A) 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 30uM Trolox+B 100uM Trolox+B 300uM Trolox+B * * # # # # # # 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 30uM Trolox+B 100uM Trolox+B 300uM Trolox+B CTL 1mM BSO 30uM Trolox+B 100uM Trolox+B 300uM Trolox+B * * # # # # # # (B) 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 300uM NAC+B 1mM NAC+B 3mM NAC+B * 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 300uM NAC+H 1mM NAC+H 3mM NAC+H * * # # # # # # 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 300uM NAC+B 1mM NAC+B 3mM NAC+B * 0 20 40 60 80 100 120 24h 36h CTL 1mM BSO 300uM NAC+B 1mM NAC+B 3mM NAC+B CTL 1mM BSO 300uM NAC+B 1mM NAC+B 3mM NAC+B * 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 300uM NAC+H 1mM NAC+H 3mM NAC+H * * # # # # # # 0 20 40 60 80 100 120 12h 24h L D H re le a se (% o f m a x im a l d ea th ) CTL 50uM H2O2 300uM NAC+H 1mM NAC+H 3mM NAC+H * * # # # # # #
Fig. 2. Effects of antioxidants in oxidative stress-induced neuronal death (A) Primary
cortical neurons were treated with antioxidants 30 min prior to 50 μM hydrogen peroxide (H2O2): trolox (30-300 μM), and NAC (300 μM -3 mM). (B) Primary cortical neurons
were treated with antioxidants 30 min prior to 1mM BSO. Data are representative of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
C. AG490 decreased the phosphorylation of JAK2 against oxidative stress
Activation of the JAK/STAT pathway is known to be restricted to certain oxidative stress stimuli as it is induced by peroxide but not by other types of reactive oxygen species, such as superoxide (Simon et al. 1998).
To elucidate the involvement of Janus kinases 2 (JAK2) signaling pathway in the neuroprotective effect of AG490, we performed western blotting. Cortical neurons were treated for various times (5, 15, 30, and 60 min) with H2O2 and BSO. Tyrosine
phosphorylation of JAK2 was increased 5min after stimuli (Fig. 3.). At that time point, AG490 reversed the oxidative stress-induced phosphorylation of JAK2. These results suggested that JAK2 activated by certain oxidants was involved in oxidant-induced neuronal death signaling pathway. AG490, a JAK2 inhibitor suppressed activation of JAK2 and also oxidative stress-induced neuronal death.
5’CTL H AG+H 15’CTL H AG+H 30’CTL H AG+H 60’ CTL H AG+H p-JAK2 JAK2 actin (A) CTL H AG + H p -J A K 2 /J A K 2 ra ti o 0.8 0.9 1.0 1.1 1.2
5’CTL H AG+H 15’CTL H AG+H 30’CTL H AG+H 60’ CTL H AG+H
p-JAK2 JAK2 actin (A) CTL H AG + H p -J A K 2 /J A K 2 ra ti o 0.8 0.9 1.0 1.1 1.2 CTL H AG + H p -J A K 2 /J A K 2 ra ti o 0.8 0.9 1.0 1.1 1.2 p-JAK2 JAK2 actin
5’CTL B AG+B 15’CTL B AG+B 30’CTL B AG+B 60’ CTL B AG+B
(B) CTL B AG + B p -J A K 2 /J A K 2 r a ti o 0.98 0.99 1.00 1.01 1.02 p-JAK2 JAK2 actin
5’CTL B AG+B 15’CTL B AG+B 30’CTL B AG+B 60’ CTL B AG+B
(B) CTL B AG + B p -J A K 2 /J A K 2 r a ti o 0.98 0.99 1.00 1.01 1.02 CTL B AG + B p -J A K 2 /J A K 2 r a ti o 0.98 0.99 1.00 1.01 1.02
Fig. 3. Inhibition of oxidative stress-induced JAK2 phosphorylation by AG490
(A) Primary cortical neurons were incubated for various times (5, 15, 30, and 60 min) with 50 μM hydrogen peroxide following pre-treatment for 30 min with 10 μM AG490. Protein extracts were prepared and subjected to western blotting as described in Material and Methods. Relative amount of each phospho-JAK2 group was compared to normal JAK2 group. (B) Primary cortical neurons were incubated for various times with 1mM BSO following pre-treatment for 30 min with 10 μM AG490. Relative amount of each phospho-JAK2 group was compared to normal phospho-JAK2 group.
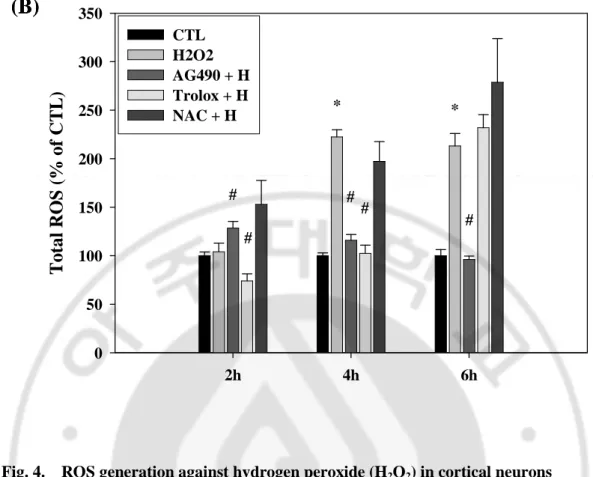
D. Oxidative stress-induced intracellular ROS production was regulated by AG490 and antioxidants
Reactive oxygen species (ROS) have been implicated as an important causative factor in cell damage, including apoptosis and necrosis. Therefore we examined whether free radicals are involved in oxidative stress-induced neurotoxicity. The intracellular ROS level was quantified with H2-DCFDA, which can be converted by ROS into DCF and then easily
visualized by strong fluorescence at around 530 nm when excited at around 480 nm.
Cortical neurons were incubated with 50µM H2O2 in the serum-free medium. The
intracellular ROS level peaked at 4h-6h after exposure to H2O2. When the pretreatment with
AG490 (10 µM), trolox (100 µM), and NAC (1mM) was carried out a half hour before H2O2
application, the change of ROS level was detected comparison with only H2O2 application.
As a result, AG490 potently blocked the production of ROS until we observed. Trolox delayed the production of intracellular ROS until 6h after H2O2 application. After 6h, trolox
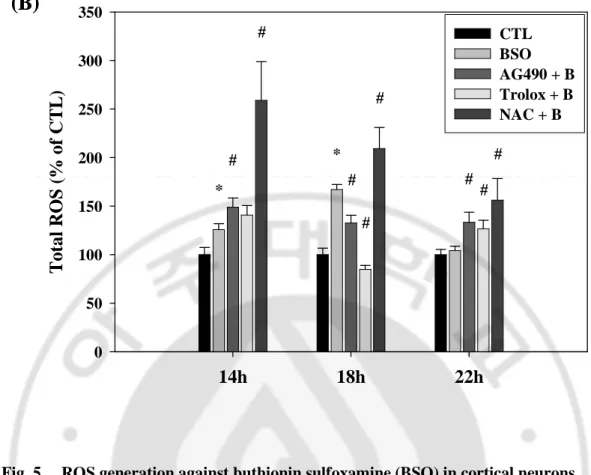
did not resist the increase of ROS level any more. In our system, cortical neurons with NAC application continuously maintained high level of the intracellular ROS at all times (Fig. 4.). When 1mM BSO was incorporated with serum-free medium in cultured neurons, the intracellular ROS production was increased at 18h. Unlike the result of H2O2 application,
ROS level against BSO was well regulated by pre-treatment of 100 µM trolox for long time. AG490 had a little decrease effect of ROS production compared to trolox. NAC induced the increase of the intracellular ROS generation by BSO in cortical neurons, same as the result of ROS generation by H2O2 (Fig. 5.).
2h
4h
6h
H2O2 50uM AG490 10uM +H2O2 Trolox 100uM +H2O2 NAC 1mM +H2O2 CTL(A)
2h
4h
6h
H2O2 50uM AG490 10uM +H2O2 Trolox 100uM +H2O2 NAC 1mM +H2O2 CTL(A)
(B)
2h 4h 6hT
o
ta
l
R
O
S
(
%
o
f
C
T
L
)
0 50 100 150 200 250 300 350 CTL H2O2 AG490 + H Trolox + H NAC + H * * # # # # #(B)
2h 4h 6hT
o
ta
l
R
O
S
(
%
o
f
C
T
L
)
0 50 100 150 200 250 300 350 CTL H2O2 AG490 + H Trolox + H NAC + H * * # # # # #Fig. 4. ROS generation against hydrogen peroxide (H2O2) in cortical neurons (A) Primary cortical neurons were incubated for 2, 4, and 6h with 50 μM hydrogen peroxide following pre-treatment for 30 min with 10 μM AG490 and antioxidants, such as trolox (100 μM), and NAC (1mM). The intracellular ROS generated by hydrogen peroxide was increased at 4h and decreased by AG490 and trolox except for NAC. Images were taken with a ZEISS fluorescence microscope. (B) Relative amount of each group compared to time control group. Data are representative of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
14h
18h
BSO 1mM AG490 10uM +BSO Trolox 100uM +BSO NAC 1mM +BSO22h
CTL(A)
14h
18h
BSO 1mM AG490 10uM +BSO Trolox 100uM +BSO NAC 1mM +BSO22h
CTL(A)
(B)
14h
18h
22h
To
ta
l
R
O
S
(
%
o
f
C
T
L
)
0 50 100 150 200 250 300 350 CTL BSO AG490 + B Trolox + B NAC + B * * # # # # # # # #(B)
14h
18h
22h
To
ta
l
R
O
S
(
%
o
f
C
T
L
)
0 50 100 150 200 250 300 350 CTL BSO AG490 + B Trolox + B NAC + B * * # # # # # # # #Fig. 5. ROS generation against buthionin sulfoxamine (BSO) in cortical neurons
(A) Primary cortical neurons were incubated for 14, 18, and 22h with 1mM buthionin sulfoxamine following pre-treatment for 30 min with 10 μM AG490 and antioxidants, such as trolox (100 μM), and NAC (1mM). The intracellular ROS generated by buthionin sulfoxamine was increased at 18h and decreased by AG490 and trolox except for NAC. Images were taken with a ZEISS fluorescence microscope. (B) Relative amount of each group compared to time control group. Data are representative of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
E. Antioxidant activities in vitro
We examined the effect of AG490 and antioxidants in oxidative stress-induced neuronal death. AG490 rather than antioxidants critically blocked cell death by oxidative stress. Antioxidant activities of AG490 itself was measured to clarify the protection mechanism against oxidative stress compared with reference antioxidants; trolox, NAC, and butylated hydroxytoluene (BHT, positive control for lipid peroxidation).
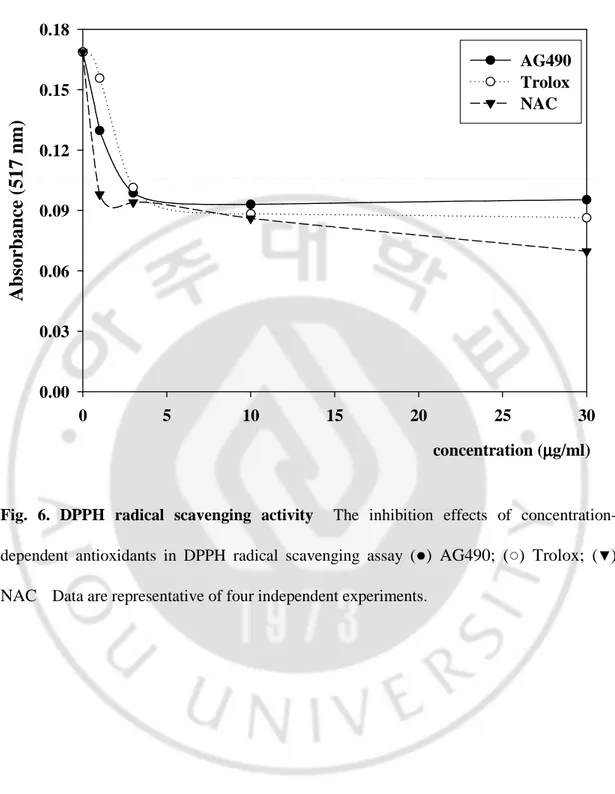
In the DPPH radical scavenging assay, the antioxidants are able to reduce the stable radical DPPH to the yellow colored diphenyl-picrylhydrazine. The method is based on the reduction of alcoholic DPPH solution in the presence of a hydrogen-donating antioxidant due to the formation of the non-radical form DPPH-H by the reaction. Fig. 6. illustrates a significant decrease in the concentration of DPPH radical due to scavenging activity of AG490 and reference antioxidants. The scavenging effect of AG490 and references on the DPPH radical decreased in the order of NAC (49.0%) > Trolox (47.6%) > AG490 (44.9%) at the concentration of 10 μg/ml.
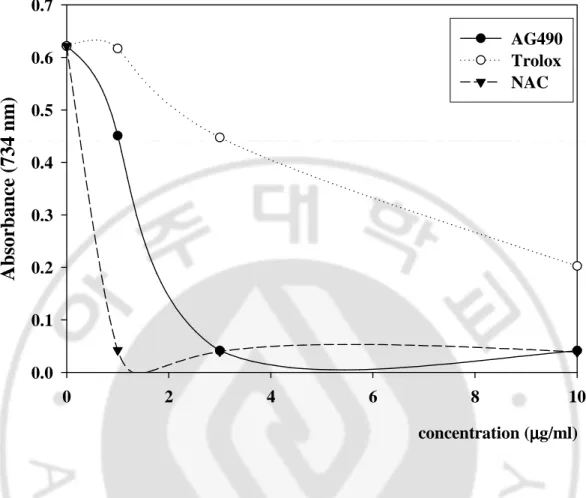
The blue/green ABTS [2, 2’-azinobis-(3-ethyl-benzothiazoline-6-sulfonic acid)] radical cation turns to transparent stable ABTS due to the reduction in the presence of antioxidant. AG490 had effective ABTS radical cation scavenging activity in a concentration-dependent manner (1-10 μg/ml). There was a significant decrease in the concentration of ABTS due to the scavenging capacity of AG490. The scavenging effect of AG490 and references on the ABTS decreased in that order: NAC (93.8%) > AG490 (93.4%) > Trolox (67.4%), at the concentration of 10 μg/ml (Fig. 7.).
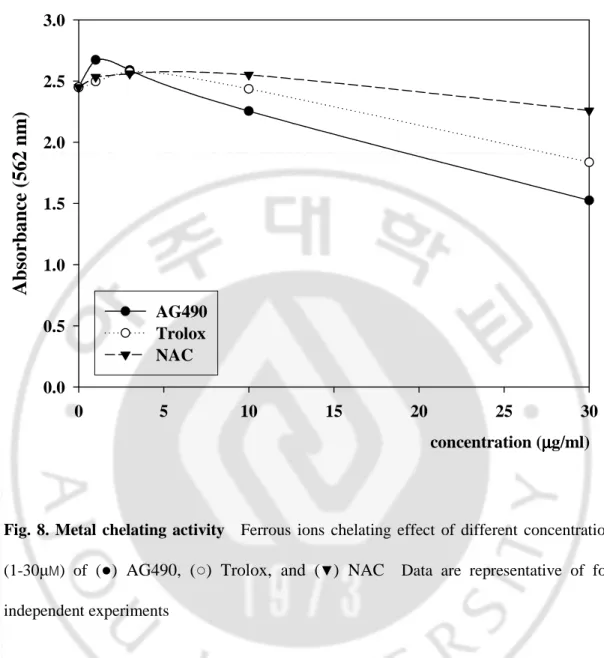
Metal chelating capacity was significant since it reduced the concentration of the catalyzing transition metal in lipid peroxidation. It was reported that chelating agents are effective as secondary antioxidants because they reduce the redox potential thereby stabilizing the oxidized form of the metal ion. AG490 exhibited about 37.7% chelation of ferrous ion at 30 μg/ml concentration. On the other hand, the percentage of metal chelating capacity of 30 μg/ml of trolox and NAC were found as 25.0% and 7.8%. The metal scavenging effect of those samples decreased in the order of AG490 > Trolox > NAC (Fig. 8.).
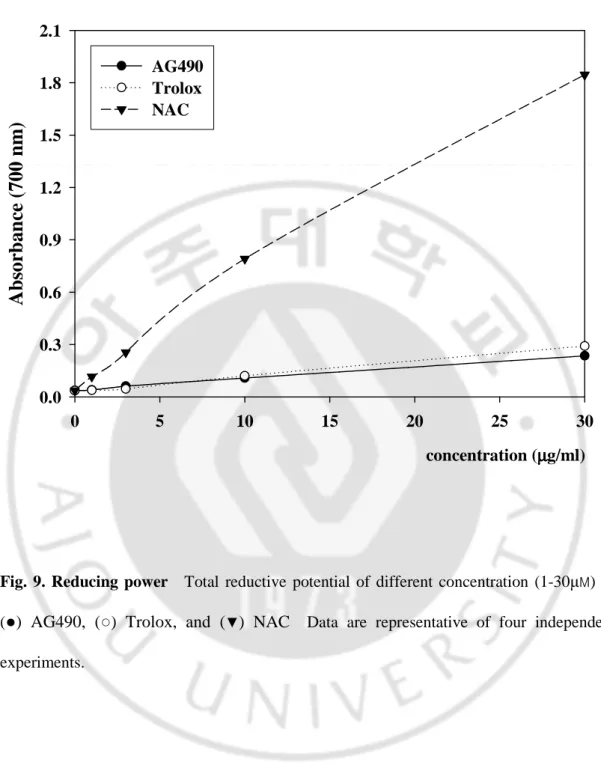
For measurements of reductive ability, the ferric ion to ferrous ion transformation was investigated in the presence of AG490 and antioxidants using the method of Oyaizu (1986). The reducing power was increased with increase of antioxidants concentrations. Reducing power of AG490 and references exhibited the following order: NAC > Trolox > AG490, at the concentration of 30 μg/ml (Fig. 9.).
C11-BODIPY 581/591 is an analogue of membrane phospholipids. The oxidation processes in membranes of living cells can be assessed by the special shift of C11-BODIPY 581/591 fluorescence upon oxidation. After incorporation of C11-BODIPY 581/591 in the cellular membrane, C11-BODIPY 581/591 changes color red to green due to oxidation of membrane. AG490 delayed the change of color of C11-BODIPY 581/591 incorporated cellular membrane. The percentage of blocking effect of lipid peroxidation of 10 μg/ml of AG490, trolox, NAC, and BHT were found as 268.8, 264.0, 98.0, and 165.9%. The protective effect of lipid peroxidation of those samples decreased in the order: AG490 > trolox > BHT > NAC, at the concentration of 10 μg/ml (Fig. 10.).
DPPH radical scavenging assay
concentration (µµµµg/ml) 0 5 10 15 20 25 30A
b
so
rb
a
n
ce
(
5
1
7
n
m
)
0.00 0.03 0.06 0.09 0.12 0.15 0.18 AG490 Trolox NACFig. 6. DPPH radical scavenging activity The inhibition effects of
concentration-dependent antioxidants in DPPH radical scavenging assay (●) AG490; (○) Trolox; (▼) NAC Data are representative of four independent experiments.
ABTS radical cation decolorization assay
concentration (µµµµg/ml) 0 2 4 6 8 10A
b
so
rb
a
n
ce
(
7
3
4
n
m
)
0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 AG490 Trolox NACFig. 7. ABTS radical cation decolorization assay The inhibition effects of
concentration-dependent antioxidants in ABTS radical cation decolorization assay. (●) AG490; (○)
Metal chelating activity
concentration (µµµµg/ml) 0 5 10 15 20 25 30A
b
so
rb
a
n
ce
(
5
6
2
n
m
)
0.0 0.5 1.0 1.5 2.0 2.5 3.0 AG490 Trolox NACFig. 8. Metal chelating activity Ferrous ions chelating effect of different concentrations
(1-30µM) of (●) AG490, (○) Trolox, and (▼) NAC Data are representative of four
Reducing power
concentration (µµµµg/ml) 0 5 10 15 20 25 30A
b
so
rb
a
n
ce
(
7
0
0
n
m
)
0.0 0.3 0.6 0.9 1.2 1.5 1.8 2.1 AG490 Trolox NACFig. 9. Reducing power Total reductive potential of different concentration (1-30µM) of
(●) AG490, (○) Trolox, and (▼) NAC Data are representative of four independent experiments.
AG490 Trolox NAC BHT re d /g re en f luo re sc en ce r a ti o (% o f C T L ) (C 1 1 -B O D IP Y 5 8 1 /5 9 1 i n te ns it y ) 0 50 100 150 200 250 CTL H2O2 1µµµµg/ml + H 3µµµµg/ml +H 10µµµµg/ml +H * * * * # # # # # # # ## #
Fig. 10. Lipid peroxidation with C-11 BODIPY 581/591 Cortical neurons were loaded
with 2μM C11-BODIPY 581/591 for 1h prior to exposure to 50 μM H2O2H. Cells were
pre-incubated with AG490 and antioxidants (1-10 μg/ml) for 1h prior to exposure to the oxidant. Intensity of C11-BODIPY 581/591 fluorescence (the ratio of the decay of red fluorescence and the increase of green fluorescence) was quantified on H2O2-induced oxidation at 4h. Red
fluorescence is visualized at around 610 nm when excited at around 580 nm. Green fluorescence is visualized at around 535 nm when excited at around 485 nm. Data are representative of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
F. AG490 increased the intracellular GSH level in cortical neurons
Glutathione (GSH) is the electron donor for reaction of peroxides in the glutathione peroxidase (GPx) and functions as a major antioxidant in tissue defense against oxidative stress, including the brain. Intracellular levels of reduced glutathione ( γ-glutamylcysteinylglycine, GSH) are maintained by glutathione reductase, a dimeric cytosolic enzyme that uses NADPH as a cofactor to catalyze the reduction of oxidized glutathione (GSSG) (Floreani et al, 1997).
A single administration of AG490 increased the ratio of GSH/GSSG, a marker of oxidative stress in cultured neurons. Four hours later, total GSH content increased 11.9 ± 2.3% with respect to basal level. The ratio of GSH/total GSH also changed to 105.2 ± 2.5% and the ratio of GSH/GSSG was 145.0% ± 19.2% (Table 1.).
Trolox itself did not influence GSH-related redox system. However, NAC decreased the ratio of GSH/total GSH and GSH/GSSG in our culture system. It is interesting that NAC is a well-known antioxidant, as a precursor of GSH.
Control AG490 Trolox NAC Total GSH GSH/Total GSH GSH/GSSG 100 111.902 ±± 2.318±± 104.439 ±± 6.199±± 92.909 ±± 6.040±± 100 105.245 ±±±± 2.458 99.101 ±±±± 1.146 98.192 ±±±± 0.571 100 145.042 ±± 19.240±± 94.462 ±±±± 8.334 86.833 ±±±± 4.040 (%) * * * *
Table 1. AG490 altered GSH/GSSG balance in cultured neurons.
AG490 and antioxidants were incubated for 4h in cortical neurons: AG490 (10 μM), NAC (1mM), Trolox (100 μM). Data are representative of six independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells)
Control AG490 Trolox NAC Total GSH GSH/Total GSH GSH/GSSG 100 111.902 ±± 2.318±± 104.439 ±± 6.199±± 92.909 ±± 6.040±± 100 105.245 ±±±± 2.458 99.101 ±±±± 1.146 98.192 ±±±± 0.571 100 145.042 ±± 19.240±± 94.462 ±±±± 8.334 86.833 ±±±± 4.040 (%) * * * *
Table 1. AG490 altered GSH/GSSG balance in cultured neurons.
AG490 and antioxidants were incubated for 4h in cortical neurons: AG490 (10 μM), NAC (1mM), Trolox (100 μM). Data are representative of six independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells)
G. Oxidative stress disturbed the mitochondrial membrane potential (∆Ψm:MMP) in cortical neurons.
Opening of mitochondrial permeability transition pore and loss of mitochondrial membrane potential are linked to oxidative cell death. Therefore we investigated whether AG490 and antioxidants prevent the mitochondrial depolarization caused by H2O2 and BSO.
Dihydrorhodamine is oxidized to rhodamine 123, which is highly fluorescent around 536nm when excited at about 500nm. Rhodamine 123 is lipophilic and positively charged, and tends to accumulate in mitochondria, held there by membrane potential. Cortical neurons remarkably began to lose mitochondrial membrane potential at 2h after exposure to H2O2
and completely abolished it at 4h. Application of AG490 (10 µM) with H2O2 was potently
blocked the reduction of mitochondrial membrane potential. We detected less abolishment of mitochondrial membrane potential in cortical neurons exposed to H2O2 with other
antioxidants: trolox (100 µM), and NAC (1mM) than abolishment of MMP in cells exposed to H2O2 itself (Fig. 11.). By 1mM BSO application, mitochondrial membrane potential was
depolarized around 22h. Unlike the result of H2O2 application, trolox potently blocked the
reduction of mitochondrial membrane potential and AG490 restored modestly. However, cortical neurons with NAC and BSO lost MMP earlier than application of BSO itself (Fig. 12.).
Trolox 100uM +H2O2 CTL
2h
4h
6h
H2O250uM AG490 10uM +H2O2 NAC 1mM +H2O2(A)
Trolox 100uM +H2O2 CTL2h
4h
6h
H2O250uM AG490 10uM +H2O2 NAC 1mM +H2O2(A)
(B)
2h
4h
6h
M
M
P
(
%
o
f
C
T
L
)
0 30 60 90 120 150 180 CTL H2O2 AG490 + H Trolox + H NAC + H * * * # # # # # # #(B)
2h
4h
6h
M
M
P
(
%
o
f
C
T
L
)
0 30 60 90 120 150 180 CTL H2O2 AG490 + H Trolox + H NAC + H * * * # # # # # # #Fig. 11. Reduction of mitochondrial membrane potential (MMP) against H2O2 in
cortical neurons (A) Primary cortical neurons were treated for 2, 4, 6h with 50 μM
hydrogen peroxide following pre-treatment for 30 min with 10 μM AG490 or antioxidants, such as trolox (100 μM), and NAC (1mM). Abolishment of MMP by hydrogen peroxide was detected at 4h and preserved by AG490 and antioxidants. MMP was measured with a ZEISS fluorescence microscope. (B) Relative amount of each group compared to time control group. Data are representative of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
14h
18h
BSO 1mM AG490 10uM +BSO Trolox 100uM +BSO NAC 1mM +BSO22h
CTL(A)
14h
18h
BSO 1mM AG490 10uM +BSO Trolox 100uM +BSO NAC 1mM +BSO22h
CTL(A)
(B)
14h
18h
22h
M
M
P
(
%
o
f
C
T
L
)
0 30 60 90 120 150 180 CTL BSO AG490 + B Trolox + B NAC + B * * * # # # # #(B)
14h
18h
22h
M
M
P
(
%
o
f
C
T
L
)
0 30 60 90 120 150 180 CTL BSO AG490 + B Trolox + B NAC + B * * * # # # # #Fig. 12. Reduction of mitochondrial membrane potential (MMP) against BSO in cortical neurons (A) Primary cortical neurons were treated for 14, 18, 22h with 1mM
BSO following pre-treatment for 30 min with 10 μM AG490 or antioxidants, such as trolox (100 μM), and NAC (1mM). Abolishment of MMP by BSO was detected at 22h and preserved by AG490 and trolox. MMP was measured with a ZEISS fluorescence microscope. (B) Relative amount of each group compared to time control group. Data are representative of four independent experiments and presented as mean ± SEM (*P<0.05, comparison with control cells, #P<0.05, comparison with oxidative stressed cells)
Ⅳ
Ⅳ
Ⅳ
Ⅳ. DISCUSSION
Here, we characterized the neuroprotective effects of tyrphostin B42 (AG490) in cultured primary cortical neurons. AG490, a JAK2 inhibitor decreased the release of lactate dehydrogenase by various oxidants in cortical neurons. AG490 significantly inhibited hydrogen peroxide (H2O2)-induced neuronal death. In the previous study, hydrogen peroxide
(H2O2) may directly activate receptors on the outside of the cell and may inactivate protein
tyrosine phosphatases (PTPs). Then, hydrogen peroxide activates intracellular kinases including JAK2 (Simon AR, et al, 1998). In this study, JAK2 was activated by 50 µM H2O2
at 5 min and AG490 treated prior to H2O2 decreased H2O2-induced JAK2 phosphorylation
(Fig. 3). These results suggested that neuroprotective mechanism of AG490 involves in JAK2 signaling pathway against oxidative stress. In transient focal cerebral ischemia, AG490 prevents the post-ischemic JAK2 and STAT3 phosphorylation and significantly decreases the infarct volume compared to vehicle group. Furthermore, intracerebral injection of siRNA
specific for STAT3 lead to suppressed STAT3 mRNA expression and phosphorylation, decreases the infract volume (Satriotomo et al, 2006). In rat astrocyte cultures, AG490 reduces oxidative stress induced by H2O2, which directly activate JAK2/STAT1 (Gorina et al,
2005). H2O2 induced serine (Ser)-727 STAT1 phosphorylation, tyrosine (Tyr)-701 STAT1
phosphorylation, and tyrosine (Tyr)-705 STAT3 phosphorylation. AG490 inhibited Tyr-701 STAT1 and Tyr-705 STAT3 phosphorylation induced by H2O2. AG490 did not prevent H2O2
-induced Ser-727 STAT1 phosphorylation as levels were significantly above control levels. Although AG490 fully inhibited STAT Tyr phosphorylation, it did not affect STAT1 Ser